15 micrograms/ml + 5 mg/ml Eye drops , solution in single
-dose container tafluprost/timolol
What Taptiqom is and what it is used for
What kind of medicine is it and how does it work?
Taptiqom eye drops contain tafluprost and timolol. Tafluprost belongs to a group of medicines called prostaglandin analogs and timolol belongs to a group of medicines called beta blockers. Tafluprost and timolol work together to lower the pressure in the eye. The caption is used when the pressure in the eye is too high.
What is your medicine for?
The caption is used to treat a type of glaucoma called open-angle glaucoma and also a condition called ocular hypertension in adults. Both of these conditions are associated with increased pressure in the eye and can ultimately affect your vision.
Tafluprost and timolol found in Taptiqom may also be approved to treat other conditions not mentioned in this product information. Ask your doctor, pharmacist, or another healthcare professional if you have any further questions, and always follow their instructions.
What you need to know before using Taptiqom
Do not use Taptiqom
- if you are allergic to tafluprost, timolol, beta-blockers, or any of the other ingredients of this medicine (listed in section 6).
- if you have or have previously had problems with breathing such as asthma, severe chronic obstructive bronchitis (severe lung disease that can cause wheezing, difficulty breathing, and/or prolonged coughing)
- if you have a slow heart rhythm, heart failure, or heart rhythm disturbances (irregular heart rhythm).
Warnings and precautions
Talk to your doctor, pharmacist, or nurse before using Taptiqom.
Talk to your doctor before using this medicine if you have or have ever had:
- coronary heart disease (symptoms may include chest pain, chest tightness, shortness of breath, or feeling of suffocation), heart failure, low blood pressure
- heart rhythm disturbances, such as slow heart rate
- breathing problems, asthma, or chronic obstructive pulmonary disease
- disease with poor blood circulation (such as Raynaud’s disease or Raynaud’s syndrome)
- diabetes, as timolol may mask signs and symptoms of low blood sugar
- an overactive thyroid, as timolol may mask signs and symptoms of thyroid disease
- allergy or anaphylactic reactions
- myasthenia gravis (a rare disease that causes muscle weakness)
- other eye diseases, such as diseases of the cornea (the clear membrane that covers the front of the eye) or a disease that requires eye surgery.
Tell your doctor if you have:
- kidney problems
- liver problems.
Please note that Taptiqom may have the following effects and some of them may be permanent:
- The caption may increase the length, thickness, color, and/or number of eyelashes and may cause abnormal hair growth on your eyelids.
- The caption can darken the skin around the eyes. Wipe off any solution that may have ended up on the skin. This reduces the risk of skin darkening.
- The caption can change the color of the iris (the colored part of your eye). If Taptiqom is used in only one eye, the color of the treated eye may be permanently different from the color of the other eye.
- The caption may cause hair growth in areas where the solution repeatedly comes into contact with the skin’s surface.
Before having an operation, tell your doctor that you are using Taptiqom, as timolol can change the effects of some medicines used for anesthesia.
Children and young people
The caption is not recommended for children or adolescents under 18 years of age as there is a lack of data on safety and efficacy in this age group.
Other medicines and Taptiqom
Tell your doctor or pharmacist if you are taking, have recently taken, or might take any other medicines.
The caption can affect or be affected by other medicines you use.
In particular, tell your doctor if you are using or intend to use:
- other eye drops for the treatment of glaucoma
- drugs to lower blood pressure et
- heart medication
- drugs for the treatment of diabetes
- quinidine (used to treat heart disease and some types of malaria )
- antidepressants called fluoxetine and paroxetine.
If you are using other medicines in the eye, wait at least 5 minutes after you have instilled Taptiqom before using the other medicine.
Contact lenses
Remove the contact lenses before using the eye drops and wait at least 15 minutes before putting the contact lenses back in.
Pregnancy, breastfeeding, and fertility
If you can become pregnant, you must use effective contraception during treatment with Taptiqom. Do not use Taptiqom if you are pregnant. You should not use Taptiqom if you are breastfeeding. Consult a doctor.
Driving ability and use of machinery
The caption may cause side effects, such as blurred vision, which may affect your ability to drive and/or use machinery. Wait to drive or use machines until you feel well and your vision is clear.
You are responsible for assessing whether you are fit to drive a motor vehicle or perform work that requires increased attention. One of the factors that can affect your ability in these respects is the use of drugs due to their effects anandamide effects. Description of these effects and side effects can be found in other sections. Read all the information in this leaflet for guidance. Discuss with your doctor or pharmacist if you are unsure.
The caption contains phosphate buffer
This medicine contains approximately 0.04 mg phosphate per drop corresponding to 1.3 mg/ml. If you have a severely damaged cornea, phosphate can in very rare cases cause cloudy spots on the cornea due to the accumulation of calcium during treatment.
How to use Taptiqom
Always use this medicine as directed by your doctor or pharmacist. Ask your doctor or pharmacist if you are unsure.
The recommended dose is 1 drop of Taptiqom in the eye or eyes once daily. Do not drip in more drops and do not use more often than prescribed by your doctor. This may make Taptiqom less effective.
Only use Taptiqom in both eyes if your doctor has prescribed it. Discard the opened container with remaining contents immediately after use.
To be used only as eye drops. Do not swallow.
Do not allow the single-dose container to touch the eye or the area around the eye. It may cause damage to the eye. The single-dose container can also become contaminated with bacteria that can cause an eye infection and lead to serious damage to the eye, including loss of vision. To avoid possible contamination of the single-dose container, the tip should not touch any surface.
Instructions for use:
When starting a new bag:
Do not use single-dose containers if the bag seal is broken. Open the bag along the dashed line. Write down the date you opened the bag in the designated date space on the bag.
Every time you use Taptiqom:
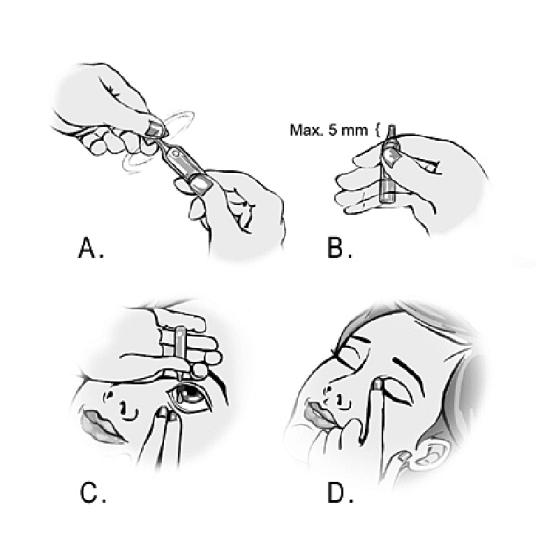
- Wash your hands.
- Take the single-dose containers out of the bag.
- Detach a single-dose container.
- Place the remaining container back into the bag and fold the edge to close the bag.
- Open the container by twisting off the tab. (Image A)
- Hold the container between your thumb and forefinger. The tip of the container must not be visible more than 5 mm above the index finger. (Image B)
- Tilt your head back or lie down. Put your hand on your forehead. The index finger should be in line with the eyebrow or rest on the bridge of the nose. Look up. Pull the lower eyelid down with the other hand. Do not allow any part of the container to come into contact with the eye or the area around the eye. Gently press the container and let a drop fall into the space between the eyelid and the eye. (Image C)
- Close the eye and press a finger on the inner corner of the eye for about two minutes. This prevents the eye drop from flowing into the tear duct. (Image D)
- Wipe off any solution that may have ended up on the skin around the eye.
If a drop ends up outside the eye, try again.
If your doctor prescribed you to use drops in both eyes, repeat steps 7 to 9 for the other eye.
The contents of one single-dose container are enough for both eyes. Discard the opened container with remaining contents immediately after use.
If you are using other medicines in the eye, wait at least 5 minutes after instilling Taptiqom before using the other medicine.
If you use too much Taptiqom
If you have taken too much Taptiqom, you may feel dizzy or have a headache, heart symptoms, or breathing problems. If you have ingested too much medicine or if, for example, a child has ingested the medicine by mistake, contact a doctor or hospital for an assessment of the risk and advice.
Consult a doctor if the medicine is accidentally swallowed.
If you forget to use Taptiqom
If you forget to use Taptiqom, drop in a single drop as soon as you remember, then go back to your routine. However, if it is almost time for the next dose, you can skip the missed dose. Do not take a double dose to make up for a missed dose.
If you stop using Taptiqom
Do not stop using Taptiqom without asking your doctor. If you stop using Taptiqom, the pressure in the eye will increase again. This can cause permanent damage to the eye.
If you have any further questions about this medicine, ask your doctor, pharmacist, or nurse.
Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. Most side effects are not serious.
You can usually continue to use the drops unless the side effects are severe. If you are worried, talk to your doctor or pharmacist.
The following are known side effects when using Taptiqom:
Common side effects are
The following side effects may occur in up to 1 in 10 users:
Eyes
Itching in the eye. Eye irritation. Eye pain. Redness in the eye. Change in length, thickness, and number of eyelashes. The sensation of a foreign body in the eye. Discoloration of the eyelashes. Light sensitivity. Blurred vision.
Less common side effects are
The following side effects may occur in up to 1 in 100 users:
Central and peripheral nervous system
Headache.
Eyes
Dry eyes. Redness of the eyelids. Small spot-like inflamed areas on the surface of the eye. Watery eyes. Swollen eyelids. Tired eyes. Inflammation of the eyelids. Inflammation inside the eye. Eye discomfort. Eye allergy. Inflammation in the eye. Abnormal sensation in the eye.
The following side effects have been seen when using the medicines Taptiqom consists of (tafluprost and timolol ) and may therefore occur when using Taptiqom:
The following side effects have been seen with tafluprost:
Eyes
Impairment in the eye’s ability to see details. Change in the color of the iris (may be permanent). Change in color of the skin around the eyes. Swelling of the surface membrane of the eye. Eye discharge. Pigmentation of the surface membrane of the eye. Follicles in the surface membrane of the eye. Sunken eyes. Iritis/uveitis ( inflammation of the colored part of the eye). Macular edema/cystic macular edema (swelling of the retina in the eye which leads to impaired vision).
The skin
Abnormal hair growth on the eyelids.
Effects on the respiratory tract
Worsened asthma, shortness of breath.
The following side effects have been seen with timolol:
The immune system
Allergic reactions, including swelling under the skin, hives, and rash. Severe sudden life-threatening allergic reaction. Itching.
Metabolism and nutrition
Low blood sugar.
Mental disorders
Depression. Difficulty sleeping. Nightmares. Memory loss. Nervousness. Hallucination.
Central and peripheral nervous system
Dizziness. Fainting. Unusual sensations (such as tingling and numbness). Increases in signs and symptoms of myasthenia gravis (muscle disease). Stroke. Decreased blood supply to the brain.
Eyes
Inflammation of the cornea. Decreased sensation in the cornea. Visual disturbances include refractive changes (sometimes due to discontinuation of miotic therapy). Drooping upper eyelids. Double vision. Blurry vision and detachment of the blood vessel-containing membrane under the retina after filtration surgery, can cause visual disturbances. Corneal damage.
Ears
Tinnitus (ringing in the ears).
The heart
Slow heartbeat. Chest pain. Palpitation. Edema (fluid accumulation). Changes in pulse or heart rhythm. Chronic heart failure (heart disease with shortness of breath, swollen feet, and legs due to fluid retention). A type of heart rhythm disorder. Heart attack. Heart failure.
Blood vessels
Low blood pressure. Limping. Raynaud’s phenomenon, is cold hands, and feet.
Breath
Spasm in the airways (preferably in people with the existing disease). Difficulty breathing. Cough.
Gastrointestinal tract
Nausea. Indigestion. Diarrhea. Dry mouth. Taste changes. Abdominal pains. Vomiting.
The skin
Hair loss. Skin rash with a white to silvery appearance (psoriasis-like rash) or worsening psoriasis. Rash.
Muscles and bones
Muscle pain is not caused by exercise. Joint pain.
Reproductive organs and mammary gland
Peyronie’s disease (which can cause bending of the penis). Sexual Dysfunction. Decreased libido.
General symptoms
Muscle weakness/fatigue. Thirst.
How to store Taptiqom
Keep this medicine out of the sight and reach of children.
Use before the expiry date which is stated on the single-dose container, sachet, and outer carton after “EXP”. The expiration date is the last day of the specified month.
Store unopened foil bags in a refrigerator (2 °C–8 °C). Do not open the bag until you need to use the eye drops as unused containers in an open bag must be discarded 28 days after the bag is first opened.
After opening the foil bag:
- Store the single-dose container in the original foil bag. Light sensitive.
- Store at a maximum of 25 °C.
- Discard unused single-dose containers 28 days after the date the foil pouch was first opened.
- Discard opened single-dose container with any remaining solution immediately after use.
Medicines must not be thrown into the drain or among the household waste. Ask the pharmacist how to dispose of medicines that are no longer used. These measures will help to protect the environment.
Contents of the packaging and other information
Contents declaration
- The active substances are tafluprost and timolol. 1 ml of solution contains 15 micrograms of tafluprost and 5 mg of timolol.
- Other ingredients are glycerol, disodium phosphate dodecahydrate, disodium edetate, polysorbate 80, hydrochloric acid, and/or sodium hydroxide (to adjust pH ), and water for injections.
Appearance and package sizes of the medicine
Caption is a clear, colorless liquid (solution) supplied in single-dose plastic containers, each containing 0.3 ml of solution. Ten single-dose containers are provided in a bag. Caption is supplied in packs containing 30 or 90 single-dose containers.
Not all pack sizes may be marketed.
Marketing Authorisation Holder
Santen Oy
Niittyhaankatu 20
33720 Tampere
Finland
Manufacturer
Santen Oy
Kelloportinkatu 1
33100 Tampere
Finland
This medicine is approved in the European Economic Area and the United Kingdom (Northern Ireland) under the name Taptiqom:
Austria, Belgium, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Latvia, Lithuania, Luxembourg, Malta, Netherlands, Norway, Poland, Portugal, Romania, Slovakia, Slovenia, Spain, Sweden, United Kingdom (Northern Ireland)
Lozada: Italy