50 micrograms / ml + 5 mg / ml eye drops , solution
latanoprost / timolol
What Latiotim is and what it is used for
Latiotim contains two medicines: latanoprost and timolol . Latanoprost belongs to the group of drugs prostaglandin analogues. Timolol belongs to the group of medicines beta-blockers. Latanoprost increases the natural outflow of aqueous humor from the eye to the bloodstream. Timolol reduces the formation of fluid inside the eye.
Latiotim is used to reduce pressure in the eye if you have open-angle glaucoma or glaucoma. Both of these diseases cause an elevated pressure in the eye that can eventually affect vision. Your doctor will usually prescribe Latiotim when other medicines do not have enough effect.
Latanoprost and timolol contained in Latiotim may also be approved for the treatment of other conditions not mentioned in this product information. Ask your doctor, pharmacist, or other healthcare professional if you have any further questions, and always follow their instructions.
What you need to know before using Latiotim
Latiotim can be used by adult men and women (including the elderly), but is not recommended if you are under 18 years of age.
Do not use Latiotim
- if you are allergic to latanoprost or timolol , beta-blockers or any of the other ingredients of this medicine (listed in section 6)
- if you have or have previously had breathing problems such as asthma or severe chronic obstructive bronchitis (severe lung disease that may cause wheezing, difficulty breathing and / or prolonged cough)
- if you have severe heart problems or arrhythmias .
Warnings and cautions
Talk to your doctor or pharmacist before using Latiotim if you have or have ever had any of the following:
- you are going to have any type of eye surgery (including cataract surgery) or have had any type of eye surgery before
- eye problems (such as eye pain, eye irritation, eye inflammation or blurred vision)
- dry eyes
- you wear contact lenses. You can still use Latiotim, but follow the instructions for contact lens wearers in section 3
- breathing problems, asthma or chronic obstructive pulmonary disease (COPD)
- disease with poor blood circulation (such as Raynaud’s disease or Raynaud’s syndrome)
- diabetes , because timolol can mask the signs and symptoms of low blood sugar
- overactive thyroid gland because timolol can mask signs and symptoms of illness
- disorders of the heart rhythm such as slow heart rhythm
- heart failure
- coronary artery disease (symptoms may include chest pain, chest tightness, shortness of breath or suffocation)
- angina (especially Prinzmetal angina)
- low blood pressure
- severe allergic reactions that usually require hospitalization
- viral infection of the eye caused by the herpes simplex virus ( HSV ).
Before using surgery, tell your doctor that you are using Latiotim, as this medicine may affect the way some medicines are used under anesthesia.
Other medicines and Latiotim
Latiotim may affect or be affected by other medicines you are taking, including other eye drops for glaucoma.
Tell your doctor or pharmacist if you are taking, have recently taken, or might take any other medicines.
In particular, tell your doctor if you are taking or plan to take any of the following medicines:
- prostaglandin is, prostaglandin analog is or prostaglandin derivative
- antihypertensive drugs (eg beta-blockers )
- medicines used to treat high blood pressure , such as oral calcium channel blockers, guanetidine , clonidine , antiarrhythmic medicines (eg amiodarone ), digitalis glycosides or parasympatomimetics
- drugs for the treatment of diabetes
- quinidine (used to treat heart problems and certain types of malaria )
- antidepressant drugs such as fluoxetine and paroxetine
- medicines used to treat allergic anaphylactic shock (adrenaline / epinephrine).
Latiotim with food and drink
Normal meals, food, or drink do not affect when or how you should use Latiotim.
Pregnancy, breastfeeding and fertility
Do not use Latiotim if you are pregnant unless your doctor tells you it’s necessary. If you are pregnant, think you may be pregnant, or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine.
Do not use Latiotim if you are breastfeeding.
The active substances of this medicine can be transferred to breast milk. If you are breastfeeding, talk to your doctor before taking any medicine.
It has been found that latanoprost and timolol do not affect male or female fertility in animal studies.
Driving and using machines
When you use Latiotim, your vision may become cloudy for a short while. If this happens, do not drive or use any tools or machines until you see clearly.
You are responsible for assessing whether you are fit to drive a motor vehicle or perform work that requires sharpened attention. One of the factors that can affect your ability in these respects is the use of drugs due to their effects and/or side effects. Descriptions of these effects and side effects can be found in other sections. Read all the information in this leaflet for guidance. If you are not sure, talk to your doctor or pharmacist.
Latiotim contains excipients
Latiotim contains benzalkonium chloride
This medicine contains 0.20 mg of benzalkonium chloride per ml. Benzalkonium chloride can be absorbed by soft contact lenses and may discolor the contact lenses. Remove contact lenses before using the medicine and wait at least 15 minutes before re-inserting the contact lenses.
Benzalkonium chloride can be irritating to the eyes, especially if you have dry eyes or problems with the cornea (the clear membrane at the front of the eye). If you experience irritation, tingling, or pain in the eye after using the medicine, consult a doctor.
Latiotim contains phosphate
This medicine contains 6.3 mg of phosphate per ml. If you have severely damaged cornea, in very rare cases, phosphate can cause cloudy spots on the cornea due to the accumulation of calcium during treatment.
How to use Latiotim
Always use this medicine exactly as your doctor has told you. Ask your doctor or pharmacist if you are unsure.
When you open the bottle for the first time, make a note of the date on the space on the outer carton provided for this purpose. Then you know when the bottle can no longer be used.
Dose
The recommended dose is
One drop in the diseased eye (s) once daily. Dose one must not exceed one (1) drop in the diseased eye (s) once daily.
Use of Latiotim at the same time as other eye drops
If you use other eye drops besides Latiotim, wait at least 5 minutes between applying the different eye drops.
Instructions for use
Follow these instructions carefully when using Latiotim eye drops. Feel free to wash your hands before use.
Do not allow the tip of the dropper to touch the eye or the area around the eye. The tip can be contaminated by bacteria that can cause eye inflammation that can lead to serious damage to the eye, even loss of vision. To avoid contamination of the dropper bottle, do not allow the tip of the dropper bottle to touch any surfaces.
Do not use the dropper bottle if the seal around the neck of the bottle is broken before the first opening of the bottle.
2. Open the bottle by turning the screw cap until the seal is broken.
3. Tilt your head back and gently pull down the lower eyelid to form a pocket between the eyelid and the eye (Figure 1).
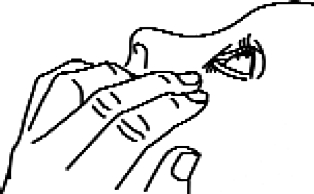
Figure 1
4. Turn the bottle so that the tip is pointing downwards and gently push (Figures 2 and 3) until one (1) drop comes in contact with the eye according to your doctor’s instructions. DO NOT TOUCH THE EYE OR THE EYLE WITH THE DROP OF THE DROP BOTTLE.
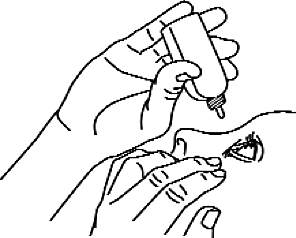
Figure 2
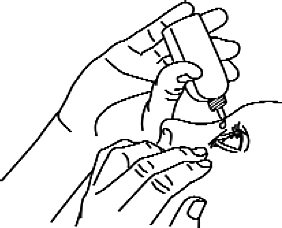
Figure 3
5. Repeat steps 3 and 4 for the other eye if your doctor prescribes it.
6. Close the eye (s) slowly after applying Latiotim and hold a finger at the corner of the eye, at the nose for 2 minutes. This prevents Latiotim from reaching other parts of the body.
7. Close the bottle immediately after use by turning the cap back firmly. Put the dropper bottle back in the original outer carton.
8. The tip of the dropper bottle is shaped to form a drop of a fixed volume; therefore, do not enlarge the hole in the tip of the dropper bottle.
Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
You can usually continue to use the drops unless the side effects are severe. If you are concerned, talk to your doctor or pharmacist. Do not stop using Latiotim without talking to your doctor.
Listed side effects known from treatment with Latiotim are listed below. The most important side effect is the risk of a gradual and permanent change in your eye color. It is also possible that Latiotim may cause serious changes in your heart function. If you notice a change in heart rhythm or heart function, contact a doctor and tell them that you have used Latiotim.
The following have been observed during treatment with Latiotim:
Very common (may affect more than 1 user in 10):
- A gradual change in eye color due to an increasing amount of brown pigment in the colored part of the eye called the iris . If you have a mottled eye color (blue-brown, gray-brown, yellow-brown or green-brown), the color change is more likely to occur than if you have monochromatic eyes (blue, gray, green or brown eyes). The change in eye color can take several years to develop. The color change may be permanent and may become more obvious if you use Latiotim in only one of the eyes. No eye problems have been identified due to. the color change. The change in eye color does not continue after discontinuation of Latiotim.
Common (may affect up to 1 in 10 people):
- eye irritation (eg a burning or burning sensation, itching , feeling of debris in the eye)
- eye pain.
Uncommon (may affect up to 1 in 100 people):
- headache
- pink / reddened eye
- dimsyn
- watery eyes
- inflammation of the eyelids
- corneal diseases
- skin rash / itching
- eye infection ( conjunctivitis )
- irritation or changes in the surface of the eye.
Other side effects are
Like other eye medicines, Latiotim (latanoprost and timolol ) are absorbed into the bloodstream. The frequency of side effects after using eye drops is lower than if the medicine is taken orally or by injection, for example.
Even if the following side effects have not been seen in connection with treatment with Latiotim, they can occur for the medicines that are included in Latiotim (latanoprost and timolol ) and thus they can occur in connection with treatment with Latiotim. The side effects listed below are those seen with the drug class beta-blockers (eg timolol ) when used to treat eye diseases:
- Development of viral infection of the eye caused by herpes simplex virus ( HSV )
- Allergic reactions affecting the whole body (with swelling under the skin that may occur on the face, arms and legs, and may block the airways and make it difficult to swallow and breathe), hives or itchy rash, local and widespread rash, itching , sudden severe life-threatening allergic reaction
- Low blood sugar level
- Dizziness
- Difficulty sleeping (insomnia), depression, nightmares, memory loss, hallucinations
- Fainting, stroke , decreased blood supply to the brain, worsening symptoms of myasthenia gravis (muscle disease), abnormal sensations (tingling or swelling of the skin) and headache.
- Swelling of the back of the eye (macular edema), fluid-filled cyst within the colored part of the eye (iris cyst), photosensitivity ( photophobia ), sunken eyes (deepening of the orbit)
- Signs and symptoms of eye irritation (eg burning, stinging, itchy feeling, tearing, red eyes), eyelid inflammation, corneal inflammation, blurred vision and discharge of the blood vessel-containing membrane under the retina after filtration surgery which may cause visual disturbance, decreased sensitivity of the cornea, dry eyes, damage to the cornea (damage to the outer layer of the eye), drooping upper eyelids (which causes the eye to be half closed), double vision
- Darkening of the skin around the eyes, change of the eyelashes and fine hair around the eye (increased number, length and thickness and darker color), changes of the direction of the eyelashes, swelling around the eye, swelling of the colored part of the eye (iritis / uveitis), scarring on the surface of the eye
- Whistling / ringing sounds in the ears ( tinnitus )
- Angina pectoris, worsening angina pectoris in patients who already have heart problems
- Slow heartbeat, chest pain, palpitations (which you feel), edema (fluid retention), changes in heart rate or heart rate, congestive heart failure (heart disease with shortness of breath, swollen feet and legs due to fluid retention), a type of disturbance of heart rhythm, heart rhythm, heart failure
- Low blood pressure , poor blood circulation that makes fingers and toes stiff and pale, cold hands and feet.
- Shortness of breath, respiratory cramps (preferably in people with pre-existing disease), difficulty breathing, cough, asthma , worsening of asthma
- Taste changes, nausea, indigestion, diarrhea, dry mouth , abdominal pain, vomiting
- Hair loss, rash with white to silvery appearance (psoriasis-like rash) or worsening psoriasis , skin rash
- Joint pain, muscle pain that is not caused by exercise, muscle weakness, fatigue
- Impaired sexual function, decreased sex drive.
In very rare cases, some patients with severe damage to the clear membrane of the front of the eye (cornea) have developed cloudy spots on the cornea due to calcium storage during treatment.
How to store Latiotim
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the label/carton after EXP. or EXP. The expiration date is the last day of the specified month.
Storage instructions before first opening:
Store in a refrigerator (2 ° C – 8 ° C).
Store in the original package. Sensitive to light.
Storage instructions after first opening:
Do not store above 25 ° C. Store in the original package. Sensitive to light.
Shelf life after opening:
28 days.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
Contents of the pack and other information
Content declaration
The active substances are latanoprost and timolol (as maleate).
One ml contains 50 micrograms latanoprost and 5 mg timolol (in the form of maleate).
The other ingredients are benzalkonium chloride, sodium dihydrogen phosphate dihydrate, anhydrous disodium phosphate, sodium chloride, sodium hydroxide / hydrochloric acid (for pH adjustment), water for injections.
What the medicine looks like and contents of the pack
Eye drops, solution
Colorless solution.
This medicine is available in clear plastic bottle with white screw cap.
Each bottle contains 2.5 ml of eye drops .
fo rpackningsstorlekar
1 x 2.5 ml, 2 x 2.5 ml, 3 x 2.5 ml, 4 x 2.5 ml, 5 x 2.5 ml, 6 x 2.5 ml, 7 x 2.5 ml, 8 x 2.5 ml, 9 x 2.5 ml, 10 x 2.5 ml and 12 x 2.5 ml
Not all pack sizes may be marketed.
Marketing Authorization Holder and Manufacturer
Marketing Authorisation Holder
Sandoz A / S, Edvard Thomsens Vej 14, 2300 Copenhagen S, Denmark
Manufacturer
Aeropharm GmbH, Francois-Mitterrand-Allee 1, 07407 Rudolstadt, Germany
or
Salutas Pharma GmbH, Otto-von-Guericke-Allee 1, 39179 Barleben, Germany
or
Lek Pharmaceuticals dd, Verovskova 57, 1526 Ljubljana, Slovenia