0.3 mg / ml + 5 mg / ml eye drops , solution
bimatoprost / timolol
1. What GANFORT is and what it is used for
GANFORT contains two different active substances (bimatoprost and timolol ), both of which lower the pressure in the eye. Bimatoprost belongs to a group of medicines called prostanoids, it is a prostaglandin analogue . Timolol belongs to a group of medicines called beta-blockers.
Your eyes contain a clear, watery liquid that nourishes the inside of the eye. Fluid is continuously drained out of the eye and new fluid is formed to replace it. If the fluid cannot be drained out of the eye fast enough, the pressure in the eye builds up and can eventually damage your vision. (This is a disease called glaucoma .) GANFORT works by reducing fluid production and increasing fluid drainage. In this way, the pressure in the eye is reduced.
GANFORT eye drops are used to treat high pressure in the eye in adults, including the elderly. High pressure can lead to glaucoma. Your doctor will prescribe GANFORT if other eye drops that contain beta-blockers or prostaglandin analogues have not given you enough effect on your own.
2. What you need to know before using GANFORT
Do not use GANFORT eye drops, solution:
- if you are allergic to bimatoprost, timolol , beta-blockers or any of the other ingredients of this medicine (listed in section 6)
- if you have or have had breathing problems such as asthma and / or severe chronic obstructive pulmonary disease (lung disease that can lead to wheezing, difficulty breathing and / or prolonged cough) or other types of breathing problems
- if you have heart problems such as low heart rate, heart block or heart failure .
Warnings and cautions
Talk to your doctor before using GANFORT if you have or have had:
- coronary heart disease (symptoms may include chest pain or pressure over the chest, shortness of breath or suffocation), heart failure , low blood pressure
- disturbed heart rhythm such as slow heart rhythm
- respiratory problems, asthma or chronic obstructive pulmonary disease (COPD)
- poor blood circulation such as Raynaud’s disease or Raynaud’s syndrome
- overactive thyroid gland because timolol can obscure signs and symptoms of thyroid disease
- diabetes because timolol can mask the signs and symptoms of low blood sugar
- severe allergic reaction
- liver or kidney problems
- problems with the surface of the eye
- loosening of any of the various layers inside the eyeball after surgery performed to lower the pressure in the eye
- known risk factors for macular edema (swelling of the retina of the eye leading to impaired vision), e.g. cataract surgery.
If you are going to receive anesthesia or anesthesia for a surgical procedure, tell your doctor that you are taking GANFORT, as timolol may alter the effect of certain medicines used in anesthesia and anesthesia.
GANFORT can cause the eyelashes to darken and grow and also cause the skin around the eyelid to darken. The color of the iris can also get darker over time. These changes can be permanent. The change can be more noticeable if only one eye is treated. GANFORT can cause hair growth in contact with the skin surface.
Children and young people
GANFORT should not be used by children and adolescents under 18 years of age.
Other drugs and GANFORT
GANFORT may affect or be affected by other medicines you are taking, including other eye drops used to treat glaucoma. Tell your doctor or pharmacist if you have recently taken or might take any other medicines. Tell your doctor if you are taking or plan to take medicines that lower your blood pressure, heart medicines, medicines that treat diabetes, quinidine (used to treat heart problems and certain types of malaria ), or medicines that treat depression called fluoxetine and paroxetine.
Pregnancy and breastfeeding
If you are pregnant or breastfeeding, think you may be pregnant, or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine. Do not use GANFORT when you are pregnant, unless your doctor specifically recommends it.
Do not use GANFORT if you are breastfeeding. Timolol may pass into breast milk. Consult your doctor before taking any medication while breastfeeding.
Driving and using machines
GANFORT may cause blurred vision in some patients. Do not drive or use machines until symptoms have resolved.
GANFORT contains excipients
GANFORT contains a preservative called benzalkonium chloride.
This medicine contains 0.15 mg of benzalkonium chloride in each 3 ml solution, equivalent to 0.05 mg/ml.
Benzalkonium chloride can be absorbed by soft contact lenses and can change the color of the contact lenses. You should remove contact lenses before using this medicine and wait at least 15 minutes before re-inserting them.
Benzalkonium chloride can also cause eye irritation, especially if you have dry eyes or problems with the cornea (the transparent layer on the front of the eye). Talk to your doctor if you experience an abnormal feeling, tingling, or pain in the eye after using this medicine.
3. How to use GANFORT
Always use this medicine exactly as your doctor or pharmacist has told you. Ask your doctor or pharmacist if you are unsure.
The recommended dose is 1 drop once daily, either in the morning or in the evening, in each eye to be treated. It should be taken at the same time each day.
Instructions for use
Do not use the bottle if the seal on the neck of the bottle is broken before you start using it.
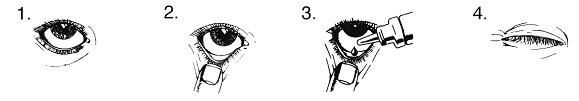
- Wash your hands. Tilt your head back and look at the ceiling.
- Carefully pull down the lower eyelid to form a small pocket.
- Turn the bottle upside down and press until there is a drop in each eye that needs treatment.
- Release the lower eyelid and close your eyes.
- Continue to close your eyes and press with your fingertip against the corner of the closed eye (where the eye meets the nose) and hold for 2 minutes. This helps prevent GANFORT from spreading to the rest of the body.
If the drop does not get in the eye, try again.
To prevent contamination, avoid the tip of the bottle coming into contact with the eye or anything else. Replace the screw cap and close the bottle immediately after use.
If you are using GANFORT with any other ophthalmic medicine, wait at least 5 minutes between the application of GANFORT and the other medicine. Use any lubricating eye drop or eye gel last.
If you use more GANFORT than you should
If you use more GANFORT than you should, it is unlikely to seriously harm you. Apply the next dose at the usual time. If you have any further questions, ask your doctor or pharmacist.
If you forget to use GANFORT
If you forget to take GANFORT, drip a drop as soon as you remember and then return to your normal routine. Do not take a double dose to make up for a forgotten dose.
If you stop using GANFORT
GANFORT should be used every day for the medicine to work as intended.
If you have any further questions on the use of this product, ask your doctor, pharmacist, or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
You can usually continue to use the drops unless the side effects are severe. If you are worried, talk to your doctor or pharmacist. Do not stop treatment with GANFORT without consulting your doctor.
The following side effects may occur with GANFORT (multiple-dose and/or single dose):
Very common side effects are
These can occur in more than 1 in 10 users
Affects the eye
Red eyes
Common side effects are
These can occur in 1 to 9 out of 100 users
Affects the eye
burning sensation, itching, tingling sensation, irritation of the conjunctiva (transparent layer in the eye), photosensitivity, eye pain, sticky eyes, dry eyes, a feeling of having something in the eye, small damage to the surface of the eye with or without inflammation, difficulty seeing clear, redness and itching of the eyelids, hair growth around the eyes, the darker skin color of the eyelids, darker skin color around the eyes, longer eyelash scars, eye irritation, watery eyes, swollen eyelids, impaired vision.
Effects on other parts of the body:
runny nose, headache.
Uncommon side effects are
These can occur in 1 to 9 out of 1,000 users
Affects the eye
abnormal sensation in the eye, inflammation of the iris, swollen conjunctiva (transparent layer in the eye), painful eyelids, tired eyes, ingrown eyelashes, darker iris color, the eyes look sunken, the eyelid moves away from the surface of the eye, darker eyelashes.
Effects on other parts of the body:
shortness of breath.
Side effects are without known frequency
Affects the eye
cystic macular edema (swelling of the retina in the eye which leads to impaired vision), eye swelling, blurred vision, discomfort in the eye.
Effects on other parts of the body:
difficulty breathing/wheezing, symptoms of an allergic reaction (swelling, red eyes, or rash), changes in taste, dizziness, lower heart rate, high blood pressure, difficulty sleeping, nightmares, asthma, hair loss, discoloration of the skin around the eyes, fatigue.
Additional side effects have been seen in patients using eye drops containing timolol or bimatoprost and may possibly occur with GANFORT. Like other eye medicines, timolol is absorbed into the bloodstream. This can cause similar side effects seen with beta-blockers given intravenously (directly into a blood vessel) and/or orally (orally). The risk of side effects after using eye drops is lower than if the drugs are taken by mouth or injected. The side effects mentioned include side effectsbimatoprost and timolol used to treat eye diseases:
- Severe allergic reactions with swelling and difficulty breathing which can be life threatening.
- Low blood sugar.
- Depression, memory loss, hallucination.
- Fainting, stroke , decreased blood flow to the brain, worsening of myasthenia gravis (worsening muscle weakness), stinging sensation.
- Decreased sensation in the surface of the eye, double vision, drooping eyelids, separation of one of the layers inside the eyeball after surgery to reduce the pressure in the eye, inflammation in the surface of the eye, bleeding in the back of the eye (bleeding in the retina), inflammation inside the eye, increased blinking.
- Heart failure , irregular or absent heartbeat, slow or fast heartbeat, excess fluid (mainly water) accumulating in the body, chest pain.
- Low blood pressure , swollen or cold hands, feet and extremities caused by the blood vessels constricting.
- Cough, worsening of asthma , worsening of chronic obstructive pulmonary disease (COPD).
- Diarrhea, abdominal pain, nausea and vomiting, digestive problems, dry mouth .
- Red scaly patches on the skin, rash.
- Muscle pain.
- Decreased sex drive, sexual dysfunction .
- Weakness.
- Elevated values on the blood tests that show how your liver works.
Other side effects reported with eye drops containing phosphate:
This medicine contains 0.38 mg of phosphates in each 3 ml solution, which corresponds to 0.95 mg/ml. If you have suffered severe damage to the front of the transparent layer on the front of the eye (cornea), phosphate can in very rare cases cause cloudy spots on the cornea due to limescale deposits during the treatment.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist, or nurse. This also applies to any side effects not mentioned in this information. You can also report side effects directly to the Medical Products Agency. By reporting side effects, you can help increase drug safety information.
5. How to store GANFORT
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the bottle and carton after EXP. The expiration date is the last day of the specified month.
No special storage instructions.
After opening the package, there is a risk of contamination of the solution, which can cause infection. You must therefore discard the bottle four weeks after opening the package for the first time, even if there are still a few drops left. To help you remember, write down the date you opened the bottle in the box on the box.
The medicine should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the packaging and other information
Content declaration
- The active substances are bimatoprost 0.3 mg / ml and timolol 5 mg / ml corresponding to timolol maleate 6.8 mg / ml.
- The other ingredients are benzalkonium chloride (as a preservative ), sodium chloride, disodium phosphate heptahydrate, citric acid monohydrate and purified water. Small amounts of hydrochloric acid or sodium hydroxide may be added to give the solution the correct pH (acidity).
What the medicine looks like and contents of the pack
GANFORT is a colorless to pale yellow, clear eye drop solution in a plastic bottle. Each package contains either 1 or 3 plastic bottles, each with a screw cap. Each bottle is approximately half full and consists of 3 ml of solution. It is enough for 4 weeks of use. Not all pack sizes may be marketed.
Marketing Authorization Holder and Manufacturer
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland
Contact the representative of the marketing authorization holder to find out more about this medicine:
| België / Belgique / Belgien /Luxembourg / LuxemburgAllergan NV / SATel: +32 (0) 2 351 24 24 | IcelandTeva Pharma Iceland ehf.Phone: +354 550 3300 |
| The NetherlandsAllergan BVTel: +32 (0) 2 351 24 24 | ItalyAllergan SpATel: + 39 06 509 562 90 |
| BulgariaAllergan Bulgaria ЕООДTel: +359 (0) 800 20 280 | |
| Czech RepublicAllergan CZ sroTel: +420 800 188 818 | LatviaAbbVie SIATel: + 371 676 05000 |
| Denmark / Norway / Finland / Finland / SwedenAllergan Norden ABTlf / Puh / Tel: + 4580884560 (DK); +47 80 01 04 97 (NO); + 358 800 115 003 (FI); +46 (0) 8 594 100 00 (SE) | LithuaniaAbbVie UABTel: + 370 5 205 3023 |
| GermanyAllergan GmbHTel: +49 69 92038 10 50 | HungaryAllergan Hungary Kft.Tel .: +36 80 100 101 |
| EestiAbbVie OÜTel. +372 6231011 | AustriaPharm-Allergan GmbHTel: +43 1 99460 6355 |
| Greece / CyprusAllergan Hellas Pharmaceuticals SATηλ: +30 210 74 73 300 | PolandAllergan Sp.z ooTel .: +48 22 256 3700 |
| SpainAllergan SATel: +34 91 807 6130 | PortugalProfarin Lda.Tel: +351 21 425 3242 |
| FranceAllergan France SASTel: +33 (0) 1 49 07 83 00 | RomaniaAllergan SRLTel: +40 21 301 53 02 |
| CroatiaAbbVie dooTel: + 385 (0) 1 5625 501 | SloveniaAbbVie Biofarmacevtska družba dooTel: +386 (1) 32 08 060 |
| Ireland / Malta / United Kingdom (Northern Ireland)Allergan Pharmaceuticals Ireland + 353 1800 931 787 (IE)+356 27780331 (MT)+44 (0) 1628 494026 (UK (NI)) | Slovak RepublicAllergan SK sroTel: + 421 800 221 223 |