20 mg / ml + 5 mg / ml eye drops , solution
(dorzolamide / timolol)
1. What Cosopt is and what it is used for
Cosopt contains two drug substances: dorzolamide and timolol .
Dorzolamide belongs to a group of medicines, so-called carbohydrase inhibitors.
• Timolol belongs to a group of drugs called beta-blockers.
These drugs lower the pressure in the eye in different ways.
COST is used to lower an elevated pressure in the eye in glaucoma ( glaucoma ) when treatment with eye drops containing only beta-blocker is not sufficient.
2. What you need to know before using Cosopt
Do not use Cosopt
- if you are allergic to dorzolamide hydrochloride, timolol maleate, or any of the other ingredients of this medicine (listed in section 6).
- if you have or have previously had respiratory problems such as asthma or severe chronic obstructive bronchitis (severe lung disease that may cause wheezing, difficulty breathing, and/or prolonged cough)
- if you have a low heart rate, heart failure, or heart rhythm disturbances (irregular heartbeat)
- if you have severe kidney disease or severe kidney problems or have previously had kidney stones
- if the acidity of your blood is elevated due to elevated chloride levels in the blood (hyperchloraemic acidosis ).
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Warnings and cautions
Talk to your doctor before using Cosopt.
Tell your doctor about any medical problems or eye problems you have or have had in the past:
- coronary heart disease (symptoms may include chest pain or pressure over the chest, shortness of breath, or suffocation), heart failure, low blood pressure
- heart rhythm disorders such as low heart rate
- respiratory distress, asthma, or chronic obstructive pulmonary disease (COPD)
- a disease that involves poor blood circulation (such as Raynaud’s disease or Raynaud’s syndrome)
- diabetes, because timolol can mask the signs and symptoms of low blood sugar
- the overactive thyroid gland, as timolol can obscure the signs and symptoms of this
Tell your doctor before an operation that you are taking Cosopt, as timolol may alter the effect of certain medicines used under anesthesia. Also tell your doctor about allergies or allergic reactions such as hives, swelling of the face, lips, tongue, and/or throat which can cause breathing and swallowing difficulties.
Also, tell your doctor if you have muscle weakness or have been diagnosed with myasthenia gravis.
If you experience irritation of any kind in the eye or have other eye problems, such as red eyes or swollen eyelids, you should contact your doctor as soon as possible.
If you suspect that Cosopt is causing an allergic reaction or hypersensitivity (eg skin rash, severe skin reaction, or redness and itching of the eye), discontinue treatment and contact a doctor immediately.
Tell your doctor if you develop an eye infection, get an eye injury, undergo eye surgery, or develop a reaction with symptoms that worsen or that you have not experienced before.
Cosopt eye drops can affect the whole body.
If you wear soft contact lenses, talk to your doctor before using this medicine.
Use in children
Experience in the treatment of infants and children with Cosopt is limited.
Use in the elderly
In studies with Cosopt, the effect was comparable in elderly and younger patients.
Use in patients with hepatic impairment
Tell your doctor if you have or have had any liver problems in the past.
Other medicines and Cosopt
Cosopt can affect or be affected by other medicines you use, including other eye drops for the treatment of glaucoma. Tell your doctor if you are taking or plan to take medicines that lower your blood pressure, heart medicine, or medicines used to treat diabetes. Tell your doctor or pharmacist if you are taking, have recently taken, or might take any other medicines. This is especially important if you:
- use medicines that lower blood pressure or to treat heart disease (such as calcium channel blockers, beta-blockers, or digoxin )
- use drugs to treat irregular heartbeats such as calcium channel blockers, beta-blockers, or digoxin
- use other eye drops that contain a beta-blocker
- uses another carbohydrate inhibitor such as acetazolamide
- uses monoamine oxidase inhibitors ( MAOIs )
- use medicines you have been given for, for example, vomiting problems (parasympathomimetics). Parasympathomimetics are a type of drug that is sometimes also used to restore normal bowel movements
- use certain strong painkillers such as morphine
- use drugs to treat diabetes
- use antidepressant drugs called fluoxetine and paroxetine
- use medicines containing sulfonamide.
- uses quinidine (a medicine used to treat heart disease and certain types of malaria ).
Pregnancy and breastfeeding
Ask your doctor or pharmacist for advice before taking any medicine.
Pregnancy
Do not use Cosopt if you are pregnant unless your doctor considers it necessary.
Breast-feeding
Do not use Cosopt if you are breast-feeding. Timolol can be excreted in your breast milk. Ask your doctor for advice before taking any medicine while you are breast-feeding.
Driving and using machines
No studies on the effects on the ability to drive and use machines have been performed. Cosopt can cause side effects such as blurred vision, which may affect your ability to drive or use machines. Wait to drive or use machines until you feel good or your vision is clear. You are responsible for assessing whether you are fit to drive a motor vehicle or perform work that requires sharpened attention. One of the factors that can affect your ability in these respects is the use of drugs due to their effects and/or side effects. Descriptions of these effects and side effects can be found in other sections. Read all the information in this leaflet for guidance. If you are not sure, talk to your doctor or pharmacist.
Cosopt contains benzalkonium chloride
If you wear soft contact lenses consult a doctor before using Cosopt (the preservative benzalkonium chloride may discolor the lenses).
3. How to use Cosopt
Always use this medicine exactly as your doctor has told you. Ask your doctor or pharmacist if you are unsure. The appropriate dose and duration of treatment are determined by your doctor.
The recommended dose is a drop morning and evening in the eye (s) to be treated.
If this medicine is used with other eye drops, the eye drops should be taken at least 10 minutes apart.
Do not change the dosage without consulting a doctor.
Make sure that the tip of the bottle does not touch the eye or the area around the eye. This can lead to contamination with bacteria, which can cause an eye infection. Such an infection can lead to serious eye damage, including loss of vision. To avoid contaminating the contents of the bottle, wash your hands and make sure that the tip of the bottle does not come into contact with any objects. If you think that the eye drops are contaminated or if you get an eye infection, contact your doctor as soon as possible to discuss whether you should continue to use the eye drops in the current bottle.
Instructions for use
Do not use the bottle if the plastic sealing strip around the neck of the bottle is missing or broken. When opening the bottle for the first time, pull off the plastic sealing strip.
Each time you use Cosopt:
- Wash your hands.
- Open the bottle. Be especially careful not to touch the tip of the bottle, the skin around the eye, or the fingers.
- Tilt your head back and hold the bottle upside down above your eye.
- Pull down the lower eyelid and lookup. Hold the bottle on the flat sides and press it lightly so that a drop falls in the space between the lower eyelid and the eye.
- Press with a finger in the corner of the eye, at the nose, or close your eyes for 2 minutes. This helps prevent the drug from entering the rest of the body.
- If your doctor has prescribed treatment for your other eye, repeat steps 3 through 5.
- Put on the cap and close the bottle tightly.
[OCUMETER PLUS bottles only]
1. Before using the bottle for the first time, make sure that the sealing strip on the front of the bottle is unbroken. A gap between the bottle and the hat is normal for an unopened bottle.
2. Wash your hands first, then pull off the sealing strip.
3. Open the bottle by unscrewing the cap, turn in the direction of the arrows. Do not pull the cap straight up from the bottle, as this may impair the function of the bottle tip.
4. Tilt your head back and pull down the lower eyelid so that a pocket is formed between the eyelid and the eye.
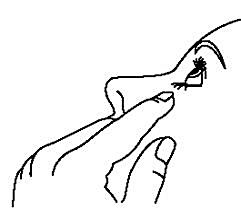
5. Turn the bottle upside down. Press lightly with your thumb or forefinger on the area where you are to press until a drop has been dropped into the eye, according to the doctor’s instructions.
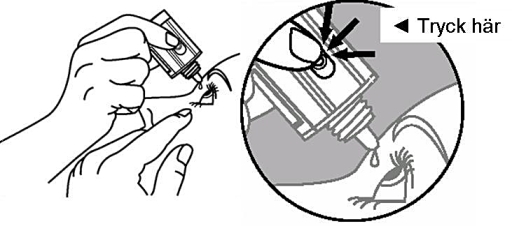
DO NOT TOUCH THE EYE OR LID WITH THE TIP OF THE BOTTLE.
6. After using Cosopt, lightly press with a finger out of the corner of your eye, your nose, or close your eyes for 2 minutes. This helps prevent the drug from entering the rest of the body.
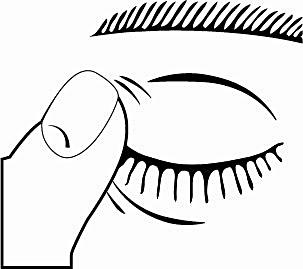
7. If it is difficult to remove droplets after opening the bottle for the first time, but the cap back on the bottle (do not tighten too hard). Remove the hat as usual by turning it according to the arrows.
8. If your doctor has prescribed treatment for your other eye, repeat steps 4 and 5.
9. Screw on the cap so that it is attached to the bottle. The arrow on the left side of the hat should be in the middle of the arrow on the left side of the bottle for the hat to fit properly. Be careful not to screw the hat too tightly as it may damage the bottle and the hat.
10. The hole in the tip of the bottle is designed to get out a single drop, therefore DO NOT make the hole in the tip of the bottle larger.
11. When you have finished using the bottle, there will be a certain amount of Cosopt left. This is normal because an extra amount of this medicine has been added. You will still receive the amount prescribed by your doctor. Do not try to get the excess medicine out of the bottle.
If you take more Cosopt than you should
If you have taken too much medicine or if, for example, a child has ingested the medicine, contact a doctor, hospital, or the Poison Information Center for risk assessment and advice. If you have dropped too many drops in the eye or have swallowed some of the contents of the container, you may, among other things, become dizzy, have difficulty breathing, or feel that your heart beats more slowly.
If you forget to take Cosopt
It is important to always take this medicine exactly as your doctor has told you. If you forget to take a dose, take it as soon as possible. However, if it is time to take the next dose, skip the missed dose and then follow your usual dosing schedule. Do not take a double dose to make up for a forgotten dose.
If you stop using Cosopt
If you want to stop treatment with the medicine, first consult your doctor. If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Serious side effects:
If you get any of the following side effects stop using this medicine and seek medical attention immediately as these may be signs of a drug reaction.
Generalized allergic reactions including swelling under the skin which may occur on areas such as the face, arms, and legs and may block the airways which may cause difficulty swallowing, shortness of breath, hives or itchy rash, local or general rash, itching, sudden severe life-threatening allergic reaction.
You can usually continue to take the drops unless the side effects are severe. If you have any further questions, ask your doctor or pharmacist. Do not stop using Cosopt without talking to a doctor.
The frequency of possible side effects listed below is defined according to the following convention:
Very common (affects more than 1 user in 10)
Common (affects less than 1 in 10 but more than 1 in 100 users)
Uncommon (affects less than 1 user in 100 but more than 1 in 1,000 users)
Rare (affects less than 1 in 1,000 but more than 1 in 10,000 users)
No known frequency (cannot be calculated from the available data)
The following side effects have been reported with Cosopt or any of its ingredients, either in clinical trials or after the drug has started to be sold:
Very common:
Burning and stinging sensation in the eyes, taste changes
Usual:
Redness in and around the eye/eyes, increased tear flow or itching in the eye/eyes, damage to the cornea (damage to the outer layer of the eye), swelling and/or irritation in or around the eye/eyes, feeling of having something in the eye, decreased sensitivity of the cornea (does not notice that something has entered the eye and does not feel pain), eye pain, dry eye, blurred vision, headache, sinusitis (it presses or feels full in the nose), nausea, weakness/fatigue.
Uncommon:
Dizziness, depression, inflammation of the iris, visual disturbances including refractive errors (in some cases due to discontinuation of treatment with so-called miotics), low heart rate, fainting, shortness of breath, indigestion, and kidney stones.
Rare:
Systemic lupus erythematosus ( SLE , an immune disorder that can cause inflammation of internal organs), tingling or numbness in the hands or feet, difficulty falling asleep, nightmares, memory loss, worsening signs and symptoms of myasthenia gravis (muscle disease), decreased sexual desire, stroke, temporary nearsightedness which may disappear when treatment is stopped, loosening of the blood vessel-containing membrane lying under the retina after filtration surgery which may cause visual disturbances, drooping eyelids (causing the eye to be half closed), double vision, scabs around the eyelids, swelling of the cornea (with symptoms of visual disturbance ), low pressure in the eye, earrings, low blood pressure , change in rhythm or heart rate,chronic heart failure (heart disease with shortness of breath, swollen feet and legs due to fluid retention), edema (fluid retention), cerebral ischemia (decreased blood supply to the brain), chest pain, severe heartbeat which may be rapid or irregular (palpitations, palpitations, palpitations) swelling or cold hands and feet as well as decreased blood circulation in the arms and legs, leg cramps and / or pain in the legs when walking (claudication), shortness of breath, respiratory failure, runny or stuffy nose, nosebleeds, contraction of the airways in the lungs, cough, throat irritation, dry mouth , diarrhea, contact dermatitis(skin inflammation), hair loss, rash with a silvery appearance (psoriasis-like rash), Peyronie’s disease (which can cause a bend of the penis), reactions of allergic type such as skin rash, hives , itching , in rare cases possible swelling of the lips, eyes and mouth, wheezing or severe skin reactions ( Stevens-Johnson syndrome , toxic epidermal necrolysis ).
Like other medicines given to your eyes, timolol is absorbed into your bloodstream. This can cause similar side effects as those seen with oral beta-blockers. The incidence of side effects when giving medicines locally in the eye is lower than when medicines are taken orally or injected, for example. Additional noted side effects are reactions observed for the class of beta-blockers in the treatment of eye diseases.
No known frequency:
Low blood sugar levels, heart failure, a type of heart rhythm disorder, abdominal pain, vomiting, muscle pain not caused by exercise, sexual dysfunction, hallucination, and feeling of debris in the eye.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist, or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly to the Medical Products Agency, www.lakemedelsverket.se. By reporting side effects, you can help increase drug safety information. Postal address
5. How to store Cosopt
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the bottle label and carton after EXP. The expiration date is the last day of the specified month.
No special temperature instructions. Store the bottle in the outer carton. Sensitive to light. The opened bottle is durable for 28 days.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the packaging and other information
Content declaration
The active substances are dorzolamide and timolol
1 ml contains 20 mg dorzolamide (as 22.26 mg dorzolamide hydrochloride) and 5 mg timolol (as 6.83 mg timolol maleate).
The other ingredients are hydroxyethylcellulose, mannitol (E421), sodium citrate (E331), sodium hydroxide (E524), and water for injections. Benzalkonium chloride is added as a preservative.
What the medicine looks like and the contents of the pack
Cosopt is a clear, colorless to almost colorless, slightly viscous solution. Cosopt is supplied in a white clear plastic bottle containing 5 ml of solution. The plastic bottle is closed with a screw cap.
The OCUMETER Plus bottle consists of a transparent plastic bottle containing 5 ml of solution. There is a sealing strip that shows that the bottle is unbroken.
Pack sizes:
1 x 5 ml (1 bottle of 5 ml)
3 x 5 ml (3 bottles of 5 ml)
6 x 5 ml (6 bottles of 5 ml)
Not all packaging may be marketed.
Marketing Authorization Holder and Manufacturer
Marketing Authorization Holder:
Santen Oy
Niittyhaankatu 20
33720 Tampere
Finland
Manufacturer:
Laboratories Merck Sharp & Dohme – Chibret
Route de Marsat, Riom
63963 Clermont-Ferrand, Cedex 9
France
or
Santen Oy
Kelloportinkatu 1
33100 Tampere
Finland
This medicinal product is authorized under the European Economic Area under the names:
In Belgium, Denmark, Finland, France, Greece, Ireland, Italy, Luxembourg, the Netherlands, Portugal, Spain, the United Kingdom, Germany, and Austria:
Cost