50 micrograms / 5 micrograms per puff, 125 micrograms / 5 micrograms per puff, 250 micrograms / 10 micrograms per puff inhalation spray , suspension
fluticasone propionate / formoterol fumarate dihydrate
1. What Flutiform is and what it is used for
Note that:
Flutiform inhalation spray, the suspension is the product name, but throughout this package leaflet, it is abbreviated to Flutiform. Sometimes it can be referred to as a specific strength.
Flutiform is an inhaler (an inhalation spray, suspension ) that contains two active ingredients:
Fluticasone propionate, which belongs to a group of medicines called steroids. Steroids help reduce swelling and inflammation in the lungs.
• Formoterol fumarate dihydrate belongs to a group of medications are known as long-acting beta2 agonists you. Long-acting beta2 agonist s are long-acting bronchodilators (LABA) that helps to keep the airways in the lungs open, making it easier for you to breathe.
Together, these two active ingredients help to improve your breathing. It is recommended that you use this medicine every day as directed by your doctor.
This medicine helps to prevent breathing problems like asthma and helps to stop shortness of breath and wheezing. However, it does not work if you already have an asthma attack, ie. if you are already wheezing and short of breath. You need to use a fast-acting bronchodilator for acute attacks such as salbutamol if this happens.
2. What you need to know before using Flutiform
Do not use Flutiform
If you are allergic to fluticasone propionate, formoterol fumarate, or any of the other ingredients of this medicine (listed in section 6).
Warnings and cautions
Talk to your doctor, pharmacist, or nurse before using this inhaler.
Before treatment with this inhaler, tell your doctor, pharmacist,t or nurse if you have:
• tuberculosis now or have had it before. Symptoms include a persistent cough, often with a bloody cough, fever, fatigue, loss of appetite, weight loss,, and night sweats.
• an infection of the lungs or chest
• heart problems such as problems with blood flow to the heart or narrowing of one of the heart valves (aortic valve), heart failure that can cause shortness of breath or swollen ankles, a condition in which the heart muscle is enlarged
( hypertrophic obstructive cardiomyopathy ), irregular heartbeat ( cardiac arrhythmia ) or if you have been told that your heart rate ( ECG ) is abnormal (prolonged QTc interval )
• an abnormal bulge of a blood vessel wall (an aneurysm )
• diabetes
• high blood pressure
• an overactive thyroid gland that may cause increased appetite, weight loss,, or sweating ( thyrotoxicosis )
• low levels of potassium in the blood, which can cause muscle weakness, twitching,, or abnormal heart rhythm ( hypokalaemia )
• poor adrenal function (if your adrenal glands do not function properly, you may experience symptoms such as headache, weakness, fatigue, abdominal pain, loss of appetite, weight loss, dizziness, very low blood pressure, diarrhea, nausea, vomiting,, or seizures) or adrenal tumor ( pheochromocytoma) )
• liver problems.
Contact a doctor if you experience blurred vision or other visual disturbances.
Tell your doctor if you are going to have surgery or are very stressed, as you may need additional steroid treatment to control your asthma.
Other medicines and Flutiform
Tell your doctor or pharmacist if you are taking, have recently taken,, or might take any other medicines, including medicines obtained without a prescription. If you use this inhaler with other medicines, the effect of this inhaler or the other medicines may change.
Tell your doctor or pharmacist if you are taking:
• medicines called beta-blockers (such as atenolol for the treatment of high blood pressure, sotalol for the treatment of irregular heartbeat, metoprolol for the treatment of fast heartbeat,, or timolol eye drops for the treatment of glaucoma )
• certain other medicines used to treat asthma or respiratory conditions (such as theophylline or aminophylline)
• medicine containing epinephrine or like substances (including other beta agonist s such as salbutamol or beta antagonist s such as atenolol, metoprolol, timolol ) Further long-acting beta2 agonist s should not be added together with this inhaler. If your asthma worsens between doses of Flutiform, use your fast-acting inhaler for immediate effect.
• medicines to treat allergic reactions ( antihistamines )
• medicines for the treatment of high blood pressure or fluid retention, which increase the amount of urine produced ( diuretics )
• medicines used to treat heart failure (such as digoxin )
• medicines to treat abnormal heart rhythms (such as quinidine, disopyramide, procainamide )
• medicines for the treatment of depressive symptoms or mental disorders such as MAO inhibitors (for example phenelzine and isocarboxazid), tricyclic antidepressants (for example amitriptyline and imipramine),, or if you have taken any of these medicines in the last two weeks
• medicines used to treat mental or mental disorders (phenothiazines or antipsychotics)
• medicines containing steroid s
• medicines for fungal infections (such as ketoconazole or itraconazole)
• some medicines may increase the effects of Flutiform and your doctor may want to check you more closely if you are taking any of these medicines (for example some medicines to treat HIV: ritonavir, atazanavir, indinavir, nelfinavir, saquinavir,, or cobicistat)
• antibiotic (such as clarithromycin, telithromycin,, or furazolidone)
• medicines to treat Parkinson’s disease ( levodopa )
• medicines for the treatment of hypothyroidism (levothyroxine)
• medicines to treat Hodgkin’s disease (procarbazine)
• medicines used to induce labor ( oxytocin ).
If you are going to have surgery under anesthesia, tell your doctor at the hospital that you are using this inhaler.
Pregnancy, breastfeeding,, and fertility
If you are pregnant or breast-feeding, think you may be pregnant,, or are planning to have a baby, ask your doctor or pharmacist for advice before using your inhaler. Your doctor will tell you if you should take this medicine.
Driving and using machines
This medicine is unlikely to affect your ability to drive or use machines.
You are responsible for assessing whether you are fit to drive a motor vehicle or perform work that requires increased attention. One of the factors that can affect your ability in these respects is the use of drugs due to their effects and/or side effects. Descriptions of these effects and side effects can be found in other sections. Read all the information in this leaflet for guidance. If you are not sure, talk to your doctor or pharmacist.
Flutiform contains ethanol (alcohol) and sodium cromoglycate
This medicine contains very small amounts of ethanol (alcohol), 1 mg per dose (puff). It also contains a very small amount of sodium cromoglycate, but patients currently taking cromoglycate (for the treatment of asthma, allergic rhinitis,, and allergic conjunctivitis ) should continue as usual.
3. How to use Flutiform
Always use this inhaler as directed by your doctor or pharmacist. Ask your doctor or pharmacist if you are unsure. You should use your inhaler regularly, ie two doses (puffs) in the morning and two doses (puffs) in the evening every day to get the most benefit from your inhaler, unless your doctor says otherwise or advises you to stop. Do not take more than the prescribed dose. Your doctor may have prescribed your inhaler for an indication other than asthma and/or a dose other than that normally prescribed and described in this leaflet. You should always use your inhaler exactly according to your doctor’s instructions. If you are not sure how much to take or how often to use your inhaler, talk to your doctor or pharmacist.
Adults, adolescents,, and children aged 5 years and older
The usual dose is two inhalations twice a day, ie two puffs in the morning and two in the evening. Your doctor will prescribe the dose required to treat your asthma.
Only adults should use the inhaler with the highest strength (Flutiform 250 micrograms / 10 micrograms).
Only adults and adolescents over 12 years of age should use the medium-strength inhaler (Flutiform 125 micrograms / 5 micrograms).
Flutiform should not be used in children under 5 years of age.
Instructions for use
Read this leaflet carefully before use and follow the instructions in the text and pictures below.
Your doctor or pharmacist will show you how to use the inhaler correctly. The medicine is contained in an aerosol container (see Figure 1), which is located inside a plastic dispenser (also called a drive unit). The drive unit also has a counter that tells you how many puffs are left after it has been prepared. This counter is color-coded. It is initially green and when there are less than 50 puffs left it turns yellow and when there are less than 30 puffs left it turns red. When it approaches zero, contact your doctor to get a replacement inhaler. Do not use the inhaler when the meter shows zero.
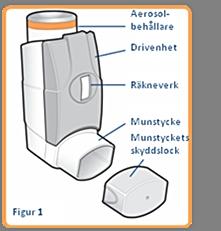
Before using your inhaler for the first time or if it has not been used for more than 3 days or if it has been exposed to frost
If your inhaler is new or has not been used for more than 3 days, it must be prepared to ensure that it works properly and gives you the right dose.
If your inhaler has been exposed to frost, it must be allowed to reach room temperature for 30 minutes,, and then it must be “prepared” to ensure that it works properly and gives the correct dose.
How to prepare the inhaler
• Remove the nozzle cover and shake the inhaler well.
• Point the nozzle away from you and release a puff by pushing down the aerosol can. This step should be performed 4 times.
The inhaler should always be shaken immediately before use.
How to use the inhaler
If you feel short of breath or wheezing while using Flutiform, continue to use Flutiform, but consult your doctor as soon as possible, as you may need further treatment. Once your asthma is well controlled, your doctor may consider it appropriate to gradually reduce the dose of one of Flutiform.
Perform steps 2 to 5 slowly.
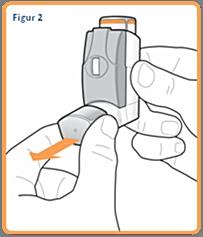
1. Remove the nozzle cover (see figure 2) and check that the inhaler is clean and free of dust.
2. Shake the inhaler immediately before releasing each puff, to ensure that the contents of the inhaler are evenly distributed.
3. Sit upright or stand. Exhale as much as feels good and as slowly and deeply as possible.
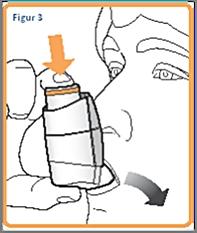
4. Hold the inhaler upright (as shown in Figure 3), place the mouthpiece in your mouth,, and close your lips around it. Hold the inhaler with your thumb (s) on the underside of the mouthpiece and your index finger (s) on the top of the inhaler. Do not bite into the nozzle.
5. Inhale slowly and deeply through your mouth and at the same time push down the aerosol can to release a puff. Continue to inhale steadily and deeply (preferably about 2-3 seconds for children or 4-5 seconds for adults).
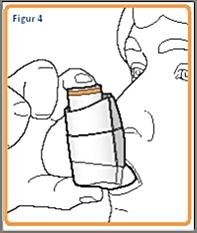
6. Hold your breath and remove the inhaler from your mouth. Keep holding your breath as long as it feels good. Finally, take the inhaler out of your mouth and exhale slowly. Do not exhale into the inhaler.
7. Before the second puff, hold the inhaler vertically and repeat steps 2 to 6.
8. Replace the nozzle cover.
You can practice in front of a mirror. If you see a “fog” coming from the top of the inhaler or around your mouth while using the inhaler, you may not have inhaled your medicine properly. Take a new dose by repeating from step 2 above.
Always rinse your mouth, gargle with water or brush your teeth after using the inhaler and spit out the residue. This can help prevent you from getting a sore mouth and throat or a hoarse voice.
If you have weak hands, it may be easier to hold the inhaler with both hands and place both index fingers on the aerosol canister and both thumbs on the bottom of the inhaler.
If you have difficulty using the inhaler, your doctor may also give you a device called the AeroChamber Plus Flow ‑ Vu breathing container, to make it easier for you to inhale your medicine into your lungs properly. Your doctor or pharmacist will tell you how to use the AeroChamber Plus Flow ‑ Vu respirator with your inhaler. AeroChamber Plus Flow ‑ Vu comes with instructions for use, care,, and cleaning instructions that you must read carefully.
Care of the inhaler
It is important that you follow these instructions carefully and that you clean your inhaler every week. To clean the inhaler, follow these steps:
• Remove the nozzle cover.
• Do not remove the aerosol canister from the drive unit.
• Wipe the inside and outside of the nozzle and drive with a clean, dry cloth or napkin.
• Replace the nozzle cover.
• Do not immerse the metal container in water.
If you use more Flutiform than you should
It is important that you take the dose indicated on the pharmacy label or prescribed by your doctor. You should not increase or decrease the dose without consulting a doctor.
If you have ingested too much medicine or if e.g. If a child has inadvertently ingested the medicine, contact a doctor, hospital,, or the Poison Information Center for risk assessment and advice.
You may have severe chest pain (angina), high or low blood pressure, headache, muscle cramps, difficulty sleeping, nervousness, dry mouth, loss of appetite, seizures, epileptic seizures,, or convulsions. You may feel shaky, dizzy, weak, tired, nauseous,, or generally ill. You may also notice changes in heart rate and you may have low levels of potassium or increased blood sugar. You may also experience symptoms such as abdominal pain, nausea, weight loss, decreased level of consciousness (which may make you feel drowsy or confused),, or low blood sugar.
If you have taken more than the prescribed dose for a long time, consult your doctor or pharmacist. This is because large doses can reduce the number of steroid hormones normally produced by your adrenal glands (see section 4).
If you forget to use Flutiform
If you forget to take a dose, take it as soon as you remember. If it is soon time for your next dose, skip the missed dose. Do not take a double dose to make up for a forgotten dose.
If you stop using Flutiform
You must use this inhaler every day according to your doctor’s instructions even if you are feeling well, as it helps to control your asthma. If you want to stop using your inhaler, talk to your doctor first. Your doctor will tell you how to do this, usually by gradually reducing the dose so that you do not trigger an asthma attack.
4. Possible side effects
Like all medicines, this inhaler can cause side effects, although not everybody gets them. Your doctor will prescribe the lowest necessary dose to control your asthma which reduces the risk of side effects.
All medicines can cause allergic reactions, although severe allergic reactions are rare. Tell your doctor immediately if you suddenly have swelling in the eyelids, face, throat, tongue or lips, rash or itching, especially if it is all over the body, symptoms such as dizziness, headache, fainting, or sudden changes in breathing patterns such as increased wheezing or shortness of breath.
Like other inhalers, your breathing may deteriorate shortly after using your inhaler. You may notice increased wheezing or shortness of breath. If this happens, stop using your Flutiform inhaler and use your fast-acting, attack-inducing inhaler instead. Contact your doctor immediately. Your doctor will assess your asthma and, if necessary, change your treatment. You should always have your attack-inducing inhaler with you.
Less common side effects are (may affect up to 1 in 100)
• Exacerbated asthma.
• Headache.
• Shaking.
• Irregular heartbeat or palpitations.
• Dizziness.
• Difficulty sleeping.
• Changed voice or hoarse voice.
Dry mouth, sore throat,, or irritated throat.
• Rash.
Rare side effects are (may affect up to 1 in 1000 users)
• An increase in the amount of sugar in the blood. If you have diabetes, you may need to check your blood sugar more often and adjust your usual diabetes treatment.
• Cod, or other fungal infections of the mouth and throat.
• Sinusitis ( sinusitis ).
• Fast heartbeat.
• Chest pain associated with heart disease.
• Muscle cramps.
• Cough or shortness of breath.
• Diarrhea.
• Indigestion.
• Changed taste.
• A feeling of dizziness or that it “spins”.
• Abnormal dreams.
• Agitation.
• Itchy skin.
• High blood pressure.
• A feeling of abnormal weakness.
• Swollen hands, ankles,, or feet.
No known frequency, (cannot be calculated from available data)
• Blurred vision.
• Sleep problems, depression or anxiety, aggression, anxiety, restlessness, nervousness, excessive arousal,, or irritability. These effects are more likely to occur in children.
The following side effects are associated with formoterol fumarate, but they have not been reported during clinical trials with this inhaler:
• Low levels of potassium in the blood, which can cause muscle weakness, twitching,, or abnormal heart rhythm.
• an abnormal heart curve that potentially leads to an abnormal heart rhythm (prolonged QTc interval )
• high levels of lactic acid in the blood.
• nausea.
• muscle pain.
Inhaled steroids can affect the normal production of steroid hormones in the body, especially if you use high doses for a long time. These effects include:
Altered bone density (thinning of skeletal bones)
Cataracts (cloudiness of the lens of the eye)
• glaucoma (increased pressure in the eye)
• bruises or skin thinning
• increased risk of getting an infection
• slower growth in children and adolescents
• a round (moon-shaped) face
• an effect on the adrenal glands (small glands located next to the kidneys), which means you may have symptoms such as weakness, fatigue, poor stress resistance, abdominal pain, loss of appetite, weight loss, headache dizziness, very low blood pressure diarrhea, nausea or vomiting or seizures.
These effects are much less likely with inhaled steroids than with steroid tablets.
Reporting of side effects ar
If you get any side effects, talk to your doctor, pharmacist,, or nurse. This also applies to side effects that are not mentioned in this information. You can also report side effects directly (see details below). By reporting side effects, you can help increase drug safety information.
The Medical Products Agency
Box 26
751 03 Uppsala
www.lakemedelsverket.se
5. How to store Flutiform
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the label, foil bag,, and carton. The expiration date is the last day of the specified month. Issue / EXP: 08-2020 means that you should not use the inhaler after the last day of that month, ie. August 2020.
Do not store above 25 ° C. Store in a cold place. Do not freeze. If the inhaler is exposed to frost, it must reach room temperature for 30 minutes and then be prepared before use (see section 3, “How to use…”). Do not use the inhaler if it has been removed from the foil bag more than 3 months ago or if the dose indicator shows “0”.
Do not expose to temperatures above 50 ° C. The aerosol container contains a compressed liquid and therefore the container must not be punctured, broken,, or burned, not even when it is apparently empty. Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the packaging and other information
Content declaration
The active substances are fluticasone propionate and formoterol fumarate dihydrate. There is inhaler er with three different strengths.
Flutiform 50 micrograms / 5 micrograms per puff, inhalation spray, suspension – only
Each puff (measured dose ) contains 50 micrograms of fluticasone propionate and 5 micrograms of formoterol fumarate dihydrate.
Flutiform 125 micrograms / 5 micrograms per puff, inhalation spray, suspension – only
Each puff (measured dose ) contains 125 micrograms of fluticasone propionate and 5 micrograms of formoterol fumarate dihydrate.
Flutiform 250 micrograms / 10 micrograms per puff, inhalation spray, suspension – only
Each puff (measured dose ) contains 250 micrograms of fluticasone propionate and 10 micrograms of formoterol fumarate dihydrate.
Other ingredients are:
• Sodium cromoglycate
• Ethanol
• Apaflurane HFA 227 ( propellant )
What the medicine looks like and the contents of the pack
These inhalers are small aerosol containers that contain a white to off-white liquid suspension and are equipped with a metering valve. The aerosol containers are located in gray and white plastic dispensers (drive units) with a light gray protective cap for the nozzle. Each inhaler contains 120 puffs. There is an inhaler in each package. Multipack is 3 x 1 inhaler (120 puffs).
Not all pack sizes may be marketed.
Marketing Authorization Holder and Manufacturer
Mundipharma AB
Mölndalsvägen 30B
412 63 Gothenburg
info@mundipharma.se
Manufacturer
Mundipharma DC BV
Listening 16
3832 RC Leusden
Netherlands
Bard Pharmaceuticals Ltd.
Cambridge Science Park
Milton Road
Cambridge CB4 0GW
UK