Estrokad 0.03 mg Vagitorium Estriol
1. What Is Estrokad,And What Estrokad Used For?
Estrogen belongs to a class of drugs called HRT ( Hormone Replacement Therapy, HRT ) and is for local use in the vagina.
Eestrokadis used for the local treatment of ailments such as dryness or irritation of the vagina in women after the last regular menstruation ( menopause ). The medical term for this is vaginal atrophy. Declining estrogen levels in the body cause it after menopause.
Estrogen works by replacing the estrogen that is normally produced in a woman’s ovaries. Estrogen is inserted into the vagina to release the hormone where it is needed. This can relieve the feeling of discomfort in the vagina.
Doctors should always be consulted before using Estrokad for the first time due to the difficulty of making the correct diagnosis the first time.
Estriol in Estrokad may also be approved for treating other conditions not mentioned in this product information. Ask your doctor, pharmacist, or healthcare professional for further questions, and always follow their instructions.
2. What You Need To Know Before Using Estrokad?
Medical History And Regular Check-ups:
The use of HRT involves risks that must be considered when deciding to start or continue an ongoing treatment. Consult a physician before starting a treatment or discuss whether to continue treatment.
Experience is limited for treating women whose menstruation has stopped prematurely (when the ovaries have stopped working or due to surgery). If your period has stopped prematurely, the risks of using HRT may be different. Talk to your doctor.
Before starting treatment (or resuming treatment), your doctor may ask about your and your family’s medical history. Your doctor may do a general medical and gynecological examination, including an examination of your breasts.
Once you have started treatment, you should go for regular medical check-ups at least once a year. During these check-ups, you should discuss the benefits and risks of continued treatment with your doctor.
Perform regular breast examinations according to your doctor’s recommendations. Before starting treatment with Estrokad, any infection in your vagina should be treated with appropriate medication.
Do not use Estrokad:
If any of the following applies to you. If unsure about the following, talk to your doctor before using Estrokad.
- If you have or have had breast cancer, or if it is suspected that you have it
- If you have cancer that is sensitive to estrogen s, such as cancer of the womb lining ( endometrium ), or if it is suspected that you have it
- If you have unexplained vaginal bleeding
- If you have a severe thickening of the uterine lining ( endometrial hyperplasia ) and are not treated for it
- If you have or have had a blood clot in a vein ( thrombosis ), in the legs (deep vein thrombosis ), or in the lungs ( pulmonary embolism )
- If you have a blood clotting disease (for example, protein C, protein S or antithrombin deficiency)
- If you have or have had a disease caused by blood clots in the arteries, for example, a heart attack, stroke ( stroke ), or chest pain (angina)
- If you have or have had liver disease and your liver function tests have not returned to normal
- If you have a rare blood disorder called “porphyria,” which is hereditary in some families
- If you are allergic to estriol or any of the other ingredients of this medicine (listed in section 6).
If any of the above conditions occur for the first time while taking Estrokad, stop using it immediately and contact your doctor.
Warnings And Cautions:
Talk to your doctor if you have or have had any of the following problems before starting treatment. They may recur or worsen during treatment with Estrokad. Should this occur, have more frequent check-ups with a doctor.
- Muscle nodules ( fibroids ) in the uterus
- Growth of the uterine lining outside the uterus ( endometriosis ) or a previous strong growth of the uterine lining ( endometrial hyperplasia ).
- Increased risk of getting blood clots (see “Blood clots in a vein ( thrombosis )”)
- Increased risk of developing estrogen-sensitive cancer (if, for example, your mother, sister, or grandmother has had breast cancer)
- High blood pressure
- Liver disease, such as a benign liver tumor
- Diabetes
- Gallstones
- Migraine or severe headache
- A disease of the immune system that affects many organs in the body (systemic lupus erythematosus, SLE )
- Epilepsy
- Asthma
- A disease that affects the eardrum and hearing (otosclerosis)
- A very high level of fat in the blood ( triglycerides er)
- Fluid retention in the body due to heart or kidney problems
You should contact your doctor immediately and discontinue treatment with Estrokad
if any of the following occurs:
- Any of the things mentioned in the section “Do not use Estrokad.”
- If the skin or whites of the eyes turn yellow (jaundice); it may be a symptom of liver disease
- If your blood pressure rises sharply (symptoms may be a headache, fatigue, or dizziness)
- If you are experiencing migraine-like headaches for the first time
- If you become pregnant
- If you get symptoms of a blood clot, such as:
- Painful swelling and redness of the legs
- Sudden chest pain
- For breathing difficulties and more information, see “Blood clots in a vein ( thrombosis ).”
Note: Estrokad is not a contraceptive. If it is less than 12 months since your last period, or if you are under 50, you may still need to use contraception to avoid pregnancy. Consult your doctor.
HRT and cancer
Severe thickening of the uterine lining ( endometrial hyperplasia ) and cancer of the uterine lining (endometrial cancer)
The use of HRT tablets with estrogen alone for a long time may increase the risk of developing cancer of the uterine lining (endometrium).
Whether repeated or prolonged (over a year) use of Estrokad involves a corresponding risk is unclear. However, it has been shown that the uptake of Estrokad into the blood is very low; therefore, adding progestin is unnecessary.
If you experience a breakthrough or splash bleeding, there is usually nothing to worry about, but you should contact a doctor. It may be a sign that your uterine lining has become thicker.
The following risks apply to hormone replacement therapy ( HRT ) circulating in the blood. On the other hand, estrogen is used topically in the vagina, and the uptake into the blood is very low. The conditions described below are unlikely to worsen or recur during treatment with Estrokad, but you should contact your doctor if you are concerned.
Breast cancer
Some indications using Estrokad does not increase the risk of breast cancer in women who have not had breast cancer before. It is unknown whether Estrokad can be used safely in women with breast cancer.
Check your breasts regularly. Contact a doctor if you notice changes such as:
- Indentations or pits
- Changes in the nipple
- Nodules you can see or feel.
It is also recommended that you participate in mammography examinations when called upon to do so.
Ovarian cancer ( ovarian cancer )
Ovarian cancer is rare – much rarer than breast cancer. The use of estrogen-only HRT has been associated with a slightly increased risk of ovarian cancer.
The risk of ovarian cancer varies with age. For example, an ovarian cancer diagnosis will be made on about 2 women out of 2,000 aged 50 to 54 who do not take HRT for 5 years. For women who have taken HRT for 5 years, there will be about 3 cases per 2,000 users (i.e., about 1 extra case).
How HRT affects the heart and blood circulation
Blood clots in a vein ( thrombosis )
The risk of blood clots in the veins is approximately 1.3–3 times higher for women who take HRT than those who do not, especially during the first year of treatment.
Blood clots can be serious. If a blood clot ends up in the lungs, it can cause chest pain, shortness of breath, collapse, or even lead death.
You are more likely to get a blood clot in a vein if you are older and if any of the following apply to you. Tell your doctor if any of the following apply to you:
- You have not been able to walk or stand for a long time due to a major operation, injury, or illness (see also section 3, “If you need surgery”)
- You are severely overweight ( BMI over 30 kg / m 2 )
- You have a coagulation disorder that requires long-term treatment with drugs that prevent blood clots.
- If a close relative has had a blood clot in the bone, lung, or another organ
- You have SLE (systemic lupus erythematosus)
- You have cancer.
The symptoms of a blood clot are described in the section “You should contact a doctor immediately and discontinue treatment with Estrokad.”
Comparison
For women in their 50s who do not take HRT, an average of 4–7 out of 1,000 are expected to have a blood clot in a vein over 5 years.
For women in their 50s who have taken HRT with estrogen alone for over 5 years, 5-8 out of 1,000 users are expected to have a blood clot in a vein (i.e., 1 extra case).
Heart disease (heart attack)
For women who take estrogen alone, there is no increased risk of developing heart disease.
Stroke (apoplexy)
The risk of stroke is about 1.5 times higher for those who take HRT than those who do not. The risk of stroke is age-dependent. Therefore, the number of stroke cases increases due to using HRT with increasing age.
Comparison
For women in their 50s who do not take HRT, an average of 8 out of 1,000 people is expected to have a stroke over 5 years. For women in their 50s who have taken HRT for more than 5 years, 11 out of 1,000 users are expected to have a stroke(i.e., 3 cases).
Other conditions
Using HRT does not prevent memory loss. The risk of memory loss may be slightly higher in women who start using HRT after age 65. Consult your doctor.
Other Medicines And Estrokad
Tell your doctor or pharmacist if you are taking or using, have recently taken or used, or might take or use any other medicines.
Because Estrokad is used for topical treatment of the vagina, it is unlikely that Estrokad would affect the effectiveness of other medicines. Estrogen may affect the effectiveness of other treatments given in the vagina.
If Estrokad is used at the same time as latex condoms, it can make it less stretchy and therefore reduce the safety of the condom.
Pregnancy And Breastfeeding:
Estrogen is intended for women whose menstruation has stopped. If you become pregnant, stop taking Estrokad and contact your doctor.
Driving And Using Machines:
Estrogen does not affect the ability to drive and use machines.
Estrogen Contains Butylhydroxytoluene:
Butylhydroxytoluene may cause local skin reactions (e.g., contact dermatitis) or irritation of the eyes and mucous membranes.
3. How To Use Estrokad?
- Always use this medicine as described in this leaflet or as your doctor or pharmacist has told you. Ask your doctor or pharmacist if you are unsure.
- The recommended dose is:
- During the first 3 weeks of treatment, a vomitorium (equivalent to 0.03 mg estriol) is used daily. After that, a maintenance dose of 1 auditorium twice a week is recommended.
- Seek medical attention if symptoms worsen or do not improve within 3-4 weeks.
- It is generally recommended to use medicines for menopausal symptoms for the shortest possible time and with the lowest possible dose that helps with the symptoms.
- Use
The auditorium should be inserted deep into the vagina before bedtime, preferably in the evening.
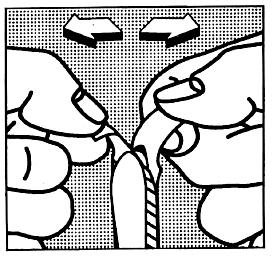
Pull the foil apart at the top when using a vomitorium until you can easily remove the auditorium.
If You Use More Estrokad, Then You Should:
Even if too many victorias were to be used at some point, there is no need to worry. However, you should consult a doctor. You may feel nauseous or vomit; some women may bleed a little from the vagina after a few days.
If you have ingested too much medicine or if, for example, a child has accidentally ingested the medicine, contact a doctor, hospital, or the Poison Information Center for risk assessment and advice.
If You Forget To Use Estrokad:
- For daily use within the first 3 weeks of treatment:
If you do not notice until the next day that you have missed a dose, skip the missed dose. Then continue with the usual dosing schedule.
- When used twice a week:
If you forget to use Estrokad on a scheduled date, make up the missed dose as soon as possible.
If You Stop Using Estrokad:
Talk to your doctor if you have questions about how long the treatment should last or discuss other options.
If you need surgery
If you have surgery, tell your surgeon that you are using Estrokad. You may need to stop using Estrokad for 4 to 6 weeks before the operation to avoid the risk of blood clots (see section 2, “Blood clots in a vein ( thrombosis )”). Ask your doctor when it is appropriate to start using Estrokad again. If you have any further questions on the use of this product, ask your doctor or pharmacist
4. Possible Estrokad Side Effects:
This medicine can cause side effects like all medicines, although not everybody gets them. The following diseases are reported more often in women who use hormone replacement therapy circulating in the blood than in non-users. These risks apply to a lesser extent to vaginally use estrogen treatments:
- Ovarian cancer ( ovarian cancer )
- Blood clots in veins in bones or lungs ( thrombosis )
- Stroke (stroke)
- Probable memory loss if HRT begins after age 65.
See section 2 for more information on these side effects. Local irritation may occur, especially at the beginning of treatment.
Common (may affect up to 1 in 10 people)
- Burning, itching, and pain in and around the vagina
- Discomfort when urinating ( dysuria ).
Uncommon (may affect up to 1 in 100 people)
- Vaginal discharge
- Discomfort in the anus and rectum.
The following side effects have been reported during treatment with HRT drugs that are taken up in the bloodstream:
- Diseases of the gallbladder
- Various skin diseases:
- Dark skin spots, especially on the face and neck, so-called “pregnancy spots” (chloasma)
- Painful reddish-purple bumps on the skin (erythema nodosum)
- Annular redness or sore rash (erythema multiforme)
5. How To Store Estrokad?
- Keep this medicine out of sight and reach of children.
- Do not use this medicine after the expiry date stated on the carton and foil after “EXP.” or “EXP.” The expiration date is the last day of the specified month.
- Do not store above 25 ° C
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines that are no longer required. These measures will help to protect the environment.
6. Contents Of The Packaging And Other Information:
Content Declaration
- The active substance is estriol.
- 1 vagitorium contains 0.03 mg estriol.
- The other ingredients are butylhydroxytoluene, glycerol mono/bis [(ZR) -12-hydroxyoctadec-9-enoate], hard fat, and macrogol cetostearyl ether.
What Does The Medicine Look Like And The Contents Of The Pack?
- Estrokad are white vagitorias.
- Estrogen is supplied in packs containing 10, 15, 20, 24, and 30 victories.
- Not all pack sizes may be marketed.
Marketing Authorization Holder And Manufacturer
DR. KADE Pharmazeutische Fabrik GmbH
Ringstrasse 2
D – 12277 Berlin
Germany
For further information on Estrokad, contact your local representative / Agent:
CampusPharma AB
Karl Gustavsgatan 1A
411 25 Gothenburg
e-mail: info@campuspharma.se
Tel: +46 31 20 50 20
This medicinal product is authorized under the European Economic Area under the names:
Denmark, Finland, Norway: Estrokad
Germany: OeKolp Ovula 0.03 mg
Hungary: Estrokad hüvelykúp
Belgium, Luxembourg: Oekolp 0.03 mg ovule / ovules / vaginal suppositories
Italy: – Atrocom 0.03 mg ovuli
United Kingdom, Ireland: – IMVAGGIS 0.03 mg pessary
Netherlands: – Estriol DR. KADE 0.03 mg ovules
Austria: – Estrokad 0.03 mg vaginal suppositories