84 mg hard capsules
eliglustat
1. What Cerdelga is and what it is used for
Cerdelga contains the active substance eliglustat and is used for long-term treatment of adult patients with Gaucher type 1 disease .
Gaucher’s type 1 disease is a rare, inherited disease in which a substance called glucosylceramide cannot be completely broken down by the body. Therefore, glucosylceramide is stored in the spleen, liver and skeleton. The storage prevents these organs from functioning normally. Cerdelga contains the active substance eliglustat, which reduces the formation of glucosylceramide and thereby prevents its storage. This in turn helps your organs to function better.
Different people break down this drug at different speeds in the body. The amount of drug in the blood can therefore vary between patients and affect how patients respond to treatment. Cerdelga is for use in patients who break down this medicine at a normal rate (also known as intermediate or fast metabolizers) or slow speed (slow metabolizers). Your doctor will determine if Cerdelga is suitable for you before you start treatment, through a simple laboratory test.
Gaucher type 1 disease is a lifelong condition and you must continue to take this medicine as your doctor prescribes to get the maximum benefit from your medicine.
What you need to know before you take Cerdelga
Do not take Cerdelga
- if you are allergic to eliglustat or any of the other ingredients of this medicine (listed in section 6).
- if you are an intermediate or fast metaboliser and are using medicines known to be strong or moderate CYP2D6 inhibitors (such as quinidine and terbinafine) in combination with strong or moderate CYP3A inhibitors (such as erythromycin and itraconazole) . The combination of these drugs affects your body’s ability to break down Cerdelga and it can lead to higher levels of the active substance in your blood (see section “Other drugs and Cerdelga” for a more comprehensive list of these drugs).
- if you are a slow metaboliser and are taking medicines called strong CYP3A inhibitors (eg itraconazole). Drugs of this type will interfere with the body’s ability to break down Cerdelga and may lead to higher levels of the active substance in the blood (see section “Other medicines and Cerdelga” for an extended list of these medicines).
- if you are a fast metabolizer and have severe hepatic impairment.
- if you are a fast metaboliser and have mild or moderate hepatic impairment while taking a strong or moderate CYP2D6 inhibitor.
Warnings and cautions
Talk to your doctor or pharmacist before taking Cerdelga if you:
- receive treatment, or should start treatment, with any of the medicines listed in the section “Other medicines and Cerdelga” .
- have had a heart attack or heart failure .
- has a low heart rate .
- have irregular or abnormal heartbeats, including a condition called long QTsyndrome.
- have some other heart problems
- taking any antiarrhythmic medicine (used to treat)irregular heartbeats) such as quinidine , amiodarone or sotalol.
- if you are a fast metabolizer and have moderate hepatic impairment.
- if you are an intermediate or slow metabolizer and have any degree of hepatic impairment.
- is an intermediate or slow metabolizer and has impaired renal function.
- is a patient with end-stage renal disease (ESRD).
Children and young people
Cerdelga has not been tested in children and adolescents under 18 years of age. Do not give this medicine to children or adolescents.
Other medicines and Cerdelga
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
Medicines that must not be taken in combination with each other and Cerdelga
Cerdelga must not be taken in combination with certain types of medicines. These medicines can affect your body’s ability to break down Cerdelga and this can lead to higher levels of Cerdelga in the blood. These drugs are known as strong or moderate CYP2D6 inhibitors and strong or moderate CYP3A inhibitors. There are many drugs like these and depending on how your body breaks down Cerdelga, the effects can differ from person to person. Talk to your doctor about these medicines before you start taking Cerdelga. Your doctor will decide which medicines you can use depending on how fast your body breaks down eliglustat.
Drugs that may increase the level of Cerdelga in the blood:
- paroxetine , fluoxetine , fluvoxamine, duloxetine, bupropion, moclobemide – antidepressants (used to treat depression)
- dronedarone, quinidine , verapamil – antiarrhythmics (used to treat irregular heartbeat)
- ciprofloxacin , clarithromycin, erythromycin, telithromycin – antibiotics (used to treat infections )
- terbinafine, itraconazole, fluconazole , posaconazole, voriconazole – antifungals(used to treat fungal infections)
- mirabegron – (used to treat overactive bladder )
- cinacalcet – calcimimetic (used by some dialysis patients and in certain cancers)
- atazanavir, darunavir, fosamprevir, indinavir, lopinavir, ritonavir, saquinavir, tipranavir – antiretroviral agents (used to treat HIV )
- kobicitat – used to improve the effect of certain HIV medicines
- aprepitant – antiemetic (against vomiting)
- diltiazem – antihypertensive agent (used to increase blood flow and lower heart rate)
- konivaptan – diuretic (used to raise low levels of sodium in the blood)
- boceprevir, telaprevir – antiviral medicine (used to treat hepatitis C)
- imatinib – cancer medicine
- amlodipine, ranolazine – used to treat angina
- cilostazol – used to treat a cramp-like pain in the legs when walking due to insufficient blood flow in the legs
- isoniazid – used to treat tuberculosis
- cimetidine, ranitidine – antacid (used to treat indigestion)
- Canadian Carrot (goldenseal) – (also known as Hydrastis canadensis ) an over-the-counter herbal preparation used to facilitate digestion.
Medicines that can reduce the level of Cerdelga in the blood:
- rifampicin, rifabutin – antibiotics (used to treat infections )
- carbamazepine, phenobarbital, phenytoin – antiepileptics (used to treat epilepsyand seizures)
- St. John’s wort – (also known as Hypericum perforatum ) an over-the-counter herbal preparation used to treat depression and other conditions
Cerdelga may increase the levels of the following types of medicines in the blood:
- dabigatran – anticoagulant (used to thin the blood)
- phenytoin – antiepileptic drug (used to treat epilepsy and seizures)
- nortriptyline, amitriptyline, imipramine, desipramine – antidepressants (used to treat depression)
- phenothiazines – antipsychotics (used to treat schizophrenia and psychosis )
- digoxin – used to treat heart failure and atrial fibrillation
- colchicine – used to treat gout
- metoprolol – used to lower blood pressure and / or heart rate
- dextromethorphan – cough medicine
- atomoxetine – used to treat ADHD
- pravastatin – used to lower cholesterol and prevent heart disease
Cerdelga with food and drink
Avoid consuming grapefruit or grapefruit juice as it may increase the level of Cerdelga in the blood.
Pregnancy, breastfeeding and fertility
If you are pregnant, think you may be pregnant or are planning to have a baby, ask your doctor for advice before taking this medicine.
Trace amounts of the active substance in this medicine have been shown to be transferred to breast milk in animals. Breast-feeding is not recommended during treatment with this medicine. Tell your doctor if you are breast-feeding.
There is no known effect on fertility at normal doses .
Driving and using machines
Cerdelga has no or negligible effect on the ability to drive and use machines.
Cerdelga contains excipients
If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicine.
How to take Cerdelga
Always take this medicine exactly as your doctor or pharmacist has told you. Ask your doctor or pharmacist if you are unsure.
If you are an intermediate or fast metabolizer:
Swallow one 84 mg capsule whole, twice daily with water. It can be taken with or without food. Take one capsule in the morning and one capsule in the evening.
If you are a slow metabolizer:
Swallow one 84 mg capsule whole, once a day with water. It can be taken with or without food. Take the capsule at the same time each day.
Do not open, crush, dissolve or chew on the capsule before swallowing. Talk to your doctor if you can not swallow the capsule whole.
Continue to take Cerdelga every day for as long as your doctor prescribes it.
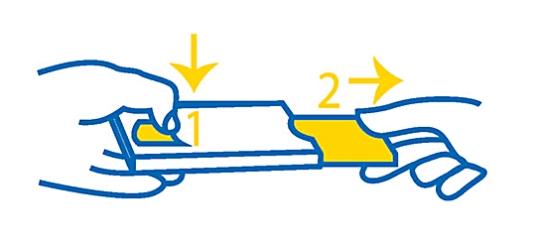
How to remove the blister / map from the case
Gently press with your thumb and a finger on one end of the case (1), gently pull out the blister / card to open the case (2).
If you take more Cerdelga than you should
If you take more capsules than you were instructed to do, contact your doctor immediately. You may have dizziness characterized by poor balance, slow heartbeat, nausea, vomiting and fainting.
If you forget to take Cerdelga
Take the next capsule at your usual time. Do not take a double dose to make up for a forgotten dose .
If you stop taking Cerdelga
Do not stop taking Cerdelga without talking to your doctor.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects , although not everybody gets them.
Common (may affect up to 1 in 10 people):
- Headache
- Dizziness
- Taste change ( dysgeusia )
- Palpitation
- Throat irritation
- Heartburn ( dyspepsia )
- Nausea
- Diarrhea
- Constipation
- Abdominal pain
- Stomach pain (pain in the upper abdomen)
- Acid reflux ( gastroesophageal reflux disease )
- Swollen stomach
- Inflammation of the stomach ( gastritis )
- Difficulty swallowing ( dysphagia )
- Vomiting
- Dry mouth
- Flatulence ( flatulence )
- Dry skin
- Hives ( urticaria )
- Joint pain ( arthralgia )
- Pain in arms, legs or back
- Fatigue (exhaustion)
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist, or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly to the Medical Products Agency, www.lakemedelsverket.se. By reporting side effects, you can help increase drug safety information.
5. How to store Cerdelga
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the carton, case and blister after “EXP”. The expiration date is the last day of the specified month.
No special storage instructions
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the packaging and other information
Content declaration
- The active substance is eliglustat (as tartrate). Each capsule contains 84 mg of eliglustat.
- Other ingredients are:
- In the capsule: microcrystalline cellulose, lactose monohydrate (see section 2 under “Cerdelga contains lactose”), hypromellose and glycerol dibehenate.
- In the capsule casing: gelatin, potassium aluminum silicate (E555), titanium dioxide (E171), yellow iron oxide (E172) and indigotine (E132).
- In the ink: shellac, black iron oxide (E172), propylene glycol and ammonia, concentrated.
What the medicine looks like and contents of the pack
Cerdelga capsules have a pearlescent blue-green, opaque top and pearlescent white, opaque base with “GZ02” printed in black on the capsule.
Pack sizes of 14 hard capsules in 1 blister card with 56 hard capsules in 4 blister cards with 14 capsules each or 196 hard capsules in 14 blister cards with 14 capsules each.
Not all pack sizes may be marketed.
Marketing Authorisation Holder
Genzyme Europe BV
Paasheuvelweg 25
1105 BP Amsterdam
Netherlands
Manufacturer
Genzyme Ireland Ltd
IDA Industrial Park
Old Kilmeaden Road
Waterford
Ireland
Contact the representative of the marketing authorization holder to find out more about this medicine:
Sanofi AB
Tel: +46 (0) 8 634 50 00