5 mg, 10 mg, 20 mg capsules, hard
oxycodone hydrochloride
What Oxycodone Actavis is and what it is used for
Oxycodone Actavis contains the active substance oxycodone hydrochloride and is a centrally acting, strong painkiller in the group opioids.
Oxycodone Actavis is used to treat severe pain that can only be alleviated sufficiently with painkillers opioid drugs.
Oxycodone hydrochloride contained in Oxycodone Actavis may also be approved for the treatment of other conditions not mentioned in this product information. Ask your doctor, pharmacist, or other healthcare professionals if you have any further questions, and always follow their instructions.
What you need to know before you use Oxikodon Actavis
Do not use Oxycodone Actavis
- if you are allergic to oxycodone hydrochloride or any of the other ingredients of this medicine (listed in section 6).
- if you have severe respiratory distress ( respiratory depression ) with too low oxygen levels in the blood ( hypoxia ) and/or too high carbon dioxide concentrations ( hypercapnia ) in the blood.
- if you have a severe chronic obstructive pulmonary disease (COPD), cor pulmonale (changes in the heart due to chronic congestion of the pulmonary circulation), or acute, severe bronchial asthma.
- if you have an intestinal obstruction (paralytic ileus ).
- if you have acute severe abdominal pain or delayed emptying of the stomach.
- if your doctor has told you not to use strong painkillers ( opioids ).
Warnings and cautions
Talk to your doctor or pharmacist before taking Oxikodon Actavis:
- if you are elderly or debilitated.
- if your lung, liver, or kidney function is severely impaired.
- if you have myxedema (a certain disease of the thyroid gland), impaired thyroid function.
- if you have impaired adrenal function (adrenal glands do not work properly) for example Addison’s disease.
- if you have an enlarged prostate ( prostatic hypertrophy ).
- if you are an alcoholic or undergoing treatment for alcohol dependence.
- if you know you are addicted to opioids.
- if you have inflammation of the pancreas ( pancreatitis ).
- in conditions with increased pressure in the brain, e.g. in case of head injury…
- if you have circulatory disorders.
- if you have colic in the bile duct and ureter.
- if you have low blood pressure or decreased blood flow to the circulatory system.
- if you have epilepsy or a tendency to seizures.
- if you are taking MAOIs (to treat depression).
- if you have recently had surgery on your abdomen or intestines.
- if you have an inflammatory bowel disease.
Tell your doctor if any of the above apply to you or if you have had any of the above before.
Oxycodone Actavis has a primary dependence potential. When used for a long time, tolerance to the effects can arise, which means that an increased dose is required to maintain pain control.
Chronic use of Oxycodone Actavis may lead to physical dependence and withdrawal symptoms may occur with abrupt discontinuation of treatment. When a patient no longer needs treatment with oxycodone hydrochloride, it is advisable to reduce the dose gradually to prevent withdrawal symptoms.
The risk of developing physical or psychological dependence is small when the drug is used as prescribed by patients suffering from chronic pain, and this risk must be weighed against the possible benefit. Discuss this with your doctor.
In rare cases, an increased sensitivity to pain, which does not respond to increased dosing, may develop. If this happens, your doctor will reduce your dose or switch to another type of opioid painkiller.
The use of Oxycodone Actavis is not recommended before surgery or 24 hours after surgery.
Oxycodone Actavis should be used with special caution in patients who are or have previously been addicted to drugs or alcohol.
Like other opioids, Oxycodone Actavis can affect the normal production of hormones in your body, such as cortisol or sex hormones. This is especially true if you are taking high doses for a long time. Symptoms may include nausea or vomiting, loss of appetite, fatigue, dizziness, altered sexual ability, changes in the menstrual cycle, or impotence. Discuss this with your doctor.
Section 4, “Possible side effects”, provides information on countermeasures to alleviate certain side effects.
Children and young people
Oxycodone Actavis has not been studied in children under 12 years of age. Safety and efficacy have not been established and therefore use in children under 12 years of age are not recommended.
Elderly patients
In elderly patients without renal and/or hepatic impairment, no dose adjustment is usually required.
Other medicines and Oxycodone Actavis
Tell your doctor or pharmacist if you are taking, have recently taken, or might take any other medicines.
If you take these capsules together with other medicines, the effect of these capsules or the other medicine may increase or decrease. The risk of side effects can also increase. Tell your doctor or pharmacist if you are taking:
- a type of medicine called a monoamine oxidase inhibitor ( MAOIs, such as moclobemide, phenelzine, isoniazid, tranylcypromine, or selegiline ), or if you have taken this type of medicine in the last two weeks (see section “Warnings and precautions”)
- medicines that help you sleep or stay calm (eg hypnotics or sedatives, including benzodiazepines )
- medicines used to treat depression (eg paroxetine or fluoxetine )
- medicines used to treat psychiatric or mental illness (such as phenothiazines or neuroleptic medicines)
- other strong painkillers ( opioids )
- muscle relaxants
- quinidine (a drug used to treat rapid heartbeat)
- cimetidine (a medicine for stomach ulcers, indigestion, or heartburn)
- medicines used to treat fungal infections (such as ketoconazole , voriconazole, itraconazole or posaconazole)
- medicines used to treat bacterial infections (such as clarithromycin, erythromycin, or telithromycin)
- a type of medicine called a protease inhibitor used to treat HIV (eg boceprevir, ritonavir, indinavir, nelfinavir, saquinavir)
- rifampicin, to treat tuberculosis
- carbamazepine (a medicine for seizures, seizures, tremors, and certain pain conditions)
- phenytoin (a medicine for seizures, seizures, and tremors)
- St. John’s wort, a traditional herbal medicine (Hypericum Perforatum)
- medicines used to treat allergies ( antihistamines ) or vomiting (antiemetics)
- drugs used to treat Parkinson’s disease
- coumarin-type anticoagulants (medicines that reduce the blood’s ability to clot)
You should also tell your doctor if you have recently been anesthetized.
Concomitant use of Oxycodone Actavis and sedatives or medicines for sleep disorders such as benzodiazepines or similar medicines increases the risk of drowsiness, difficulty breathing ( respiratory depression ), coma and may be life-threatening. Due to this, concomitant use should only be considered when other treatment options are not possible.
If your doctor prescribes Oxycodone Actavis at the same time as sedatives, the dose and treatment time should be limited by your doctor.
Tell your doctor if you are taking any sedatives and carefully follow your doctor’s dose recommendations. It may be helpful to inform friends or relatives about paying attention to the signs and symptoms described above. Contact a doctor if you experience any of these symptoms.
The risk of side effects is increased if you take antidepressant drugs (for example, citalopram, duloxetine, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, venlafaxine ). These drugs can affect or be affected by oxycodone, and you may experience symptoms such as involuntary, rhythmic muscle contractions, including in the muscles that control eye movements, sudden movements, heavy sweating, tremors, excessive reflexive movements, increased muscle tension, body temperature above 38 ° C. Contact your doctor if you experience these symptoms.
Oxycodone Actavis with food, drink, and alcohol
Oxycodone Actavis can be taken with or without food with a sufficient amount of fluid.
Grapefruit juice
Grapefruit juice can impair the breakdown of oxycodone, which increases its effectiveness. Therefore, you should avoid drinking grapefruit juice while taking Oxikodon Actavis.
Alcohol
If you drink alcohol while using Oxikodon Actavis, it may make you feel more sleepy or increase the risk of serious side effects such as shallow breathing with a risk of respiratory arrest and unconsciousness. You should not drink alcohol while using Oxikodon Actavis.
Pregnancy and breastfeeding
If you are pregnant or breastfeeding, or if you think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine.
Pregnancy
If you are pregnant, you should avoid taking Oxikodon Actavis if possible. There is insufficient experience with oxycodone treatment in pregnant women. Oxycodone passes through the placenta into the bloodstream of the fetus.
Prolonged use of oxycodone during pregnancy may cause withdrawal symptoms in the newborn. The use of oxycodone during labor can cause severe breathing problems in the newborn.
Breast-feeding
You should not take Oxycodone Actavis during the breastfeeding period as oxycodone passes into breast milk and may cause breathing problems in the baby.
Driving and using machines
Oxycodone impairs the ability to concentrate and react to such an extent that the ability to drive and use machines is affected or disappears altogether. For possible side effects affecting driving ability and concentration, see section 4 “Possible side effects”. During stable treatment, it is not necessary to ban driving in general. The treating physician must evaluate the individual situation. Discuss with your doctor if and under what circumstances you can drive.
You are responsible for assessing whether you are fit to drive a motor vehicle or perform work that requires sharpened attention. One of the factors that can affect your ability in these respects is the use of drugs due to their effects and/or side effects. The description of these effects and side effects can be found in other sections. Read all the information in this leaflet for guidance. If you are not sure, talk to your doctor or pharmacist.
Oxycodone Actavis contains sodium
This medicine contains less than 1 mmol (23 mg) sodium per capsule, ie essentially ‘sodium-free’. is next to “sodium-free”.
How to use Oxycodone Actavis
Use for children and adolescents
Always take this medicine exactly as your doctor or pharmacist has told you. Ask your doctor or pharmacist if you are unsure.
Adults and adolescents (12 years and older)
The usual starting dose is one 5 mg capsule every six hours. However, your doctor will prescribe the appropriate dose and frequency of administration to treat your pain.
If you still feel pain while taking these capsules, discuss it with your doctor.
Use
Swallow the Oxikodon Actavis capsules whole with a sufficient amount of fluid.
Oxycodone Actavis must not be taken with alcoholic beverages.
You may only take the capsules by mouth. The contents of the capsules should never be injected, as this can lead to serious and potentially fatal side effects.
Instructions for use of child-resistant blister packs:
1. Do not push the capsule directly out of the capsule compartment.
2. Separate a blister cell from the strip at the perforations.
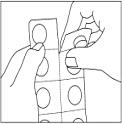
Carefully pull off the back to open the capsule compartment.
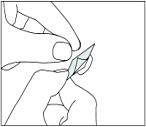
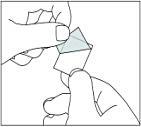
Adults with impaired renal or hepatic function
The usual starting dose is half the recommended dose for adults. Your doctor will prescribe the appropriate dose based on your clinical situation and use a more appropriate formula if available.
Use for children
Oxycodone Actavis is not recommended for children under 12 years of age.
If you use more Oxikodon Actavis then you should
If you have ingested too much medicine or if e.g. If a child has ingested the medicine by mistake, contact a doctor or hospital for risk assessment and advice. The following symptoms may occur: constricted pupils ( miosis ), decreased breathing ( respiratory depression ), muscle weakness, drowsiness, and drop in blood pressure. In severe cases, circulatory collapse, mental and motor inactivity (torpedo), unconsciousness ( coma ), slow heartbeat, and accumulation of water in the lungs (non-cardiogenic pulmonary edema ) may occur. Abuse of high doses of strong opioids such as oxycodone can be fatal. Under no circumstances should you expose yourself to situations that require a high ability to concentrate, such as driving.
If you forget to use Oxikodon Actavis
If you forget to take a dose, take the next dose as soon as you remember and then continue as usual. Never take two doses within 4 hours. Do not take a double dose to make up for a forgotten capsule.
If you stop using Oxikodon Actavis
Do not stop treatment without talking to your doctor.
When a patient no longer needs treatment with Oxycodone Actavis, it is advisable to reduce the dose gradually to prevent withdrawal symptoms.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Possible side effects
Like all medicines, Oxikodon Actavis can cause side effects, although not everybody gets them.
If you experience any of the following side effects, stop taking Oxikodon Actavis and contact your doctor immediately :
- respiratory depression, which is the most serious risk with opioids. It usually affects elderly or debilitated patients. As a result, opioids can lead to severe hypotension in some patients with an increased risk of hypotension.
- severe allergic reactions (anaphylactic shock ) which may cause hives, swelling of the face, lips, mouth, tongue, or throat, or difficulty breathing.
- reduction in pupil size
- sudden contraction of the airways which makes it difficult to breathe ( bronchospasm )
- stomach cramps
- subdued cough reflex
Other possible side effects are
Very common (may affect more than 1 user in 10):
- decreased level of consciousness (fatigue to drowsiness)
- dizziness
- headache
- constipation
- nausea
- vomiting
- itching.
Common (may affect up to 1 in 10 people):
- reduced appetite
- various psychological side effects such as
- mood swings (such as anxiety, depression).
- altered activity level (usually decreased, sometimes accompanied by fatigue, but sometimes increased with nervousness and sleep disorders)
- changed behavior (thought process disorders, confusion)
- tremors _ _
- hissing breathing, shortness of breath, hiccups
- dry mouth, stomach pain, diarrhea, indigestion ( dyspepsia )
- rash, increased sweating
- increased need to urinate
- a feeling of weakness ( asthenia ).
Uncommon (may affect up to 1 in 100 people):
- allergic reactions
- abnormal secretion of antidiuretic hormone
- lack of water in the body (dehydration)
- perceptual disorders such as personality change and seeing, hearing, or feeling things that are not there (hallucinations), decreased sex drive, restlessness, sharp mood swings, elation, drug dependence (see section 2)
- increased or decreased muscle tone, coordination difficulties, involuntary muscle contractions, seizures, especially in patients with epilepsy or tendency to seizures, increased muscle stiffness and difficulty in stretching muscles, speech difficulties, fainting, tingling or ant crawling ( paresthesia ), decreased sensitivity to touch ( hypesthesia ) migraine, altered taste experience, memory loss
- changes in tear flow, contraction of the pupils, visual disturbances
- unusually sharp hearing (hyperacusis), feeling of dizziness or dizziness ( vertigo )
- increased heart rate, awareness of heartbeat
- dilated blood vessels (vasodilation)
- breathing difficulties
- cough, sore throat, runny nose, voice changes
- difficulty swallowing, cold sores, inflammation of the gums, inflammation of the mouth ( stomatitis ), gas, belching, intestinal obstruction ( ileus )
- elevated levels of liver enzyme
- dry skin
- problems urinating
- impotence
- pain (eg chest pain), chills, too much fluid in the tissues ( edema ), malaise, physical dependence on withdrawal symptoms, drug tolerance which means an increased dose is required to maintain the effect, thirst
- injuries as a result of accidents.
Rare (may affect up to 1 in 1,000 people):
- herpes simplex (a disease that affects the skin and mucous membranes)
- lymph node disease ( lymphadenopathy )
- increased appetite
- lowered blood pressure, dizziness when walking from a lying or sitting position to standing
- bleeding gums, dark stools, stains, and damage to the teeth
- itchy rash (hives), increased photosensitivity ( photosensitivity )
- muscle spasms
- blood in the urine ( hematuria )
- changes in body weight (increased or decreased), cellulite.
Very rare (may affect up to 1 in 10,000 people):
- scaly rash (exfoliative dermatitis ).
Has been reported (occurs in an unknown number of users):
- severe hypersensitivity reaction (anaphylactic shock )
- aggressiveness
- increased pain sensitivity that does not respond to dose increase
- caries in the teeth
- pain on the right side of the abdomen, itching, and jaundice caused by inflammation of the gallbladder
- missed period ( amenorrhea )
- Long-term use of Oxycodone Actavis during pregnancy may cause life-threatening withdrawal symptoms in newborns. Symptoms to look out for in the child are e.g. irritability, hyperactivity and abnormal sleep patterns, crying, shaking, vomiting, diarrhea, and weight loss.
Prevention:
If you experience any of the side effects mentioned above, your doctor will usually take the necessary measures. Side effects in the form of constipation can be prevented through a fiber-enriched diet and increased fluid intake. If you suffer from nausea or vomiting, your doctor will prescribe a suitable medicine for this.
How to store Oxikodon Actavis
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the label or carton after EXP or EXP. The expiration date is the last day of the specified month.
Do not store above 30 ° C.
HDPE can only: Be used within 6 months after opening.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
Contents of the pack and other information
Content declaration
- The active substance is oxycodone hydrochloride. Oxycodone Actavis 5 mg: Each capsule contains 5 mg of oxycodone hydrochloride.Oxycodone Actavis 10 mg: Each capsule contains 10 mg of oxycodone hydrochloride.Oxycodone Actavis 20 mg: Each capsule contains 20 mg of oxycodone hydrochloride.
- The other ingredients are Capsule core: microcrystalline cellulose, magnesium stearate. Capsule film coating: gelatin, sodium lauryl sulfate, titanium dioxide (E171), yellow iron oxide (E172), red iron oxide (E172), indigo carmine (E132). Printing ink: shellac, black iron oxide (E172), potassium hydroxide.
What Oxycodone Actavis looks like and the contents of the pack
Oxycodone Actavis 5 mg:
Hard capsules, 14.4 mm long, dark pink, marked with “5” and a brown lid marked with “OXY”.
Oxycodone Actavis 10 mg:
Hard capsules, 14.4 mm long, white, marked with “10” and a brown lid marked with “OXY”.
Oxycodone Actavis 20 mg:
Hard capsules, 14.4 mm long, light pink, marked with “20” and a brown lid marked with “OXY”.
Pack sizes:
Blister packs: 10, 14, 20, 28, 30, 50, 56, 90, 98 and 100 capsules.
Child protection blister packs: 10, 14, 20, 28, 30, 50, 56, 90, 98 and 100 capsules.
Child protection capsule jar: 56, 98, 100 and 250 capsules.
Not all pack sizes may be marketed.
Actavis Group PTC ehf
Reykjavíkurvegur 76-78
220 Hafnarfjörður
Iceland