eye drops
caramel sodium
1. What Celluvisc is and what it is used for
Celluvisc is a clear solution in single-dose plastic containers . Each single-dosecontainer contains 0.4 ml of solution.
Celluvisc is used as a tear replacement to relieve symptoms due to dry eyes, such as chafing, burning, itching, burning sensation, and tiredness in the eyes.
You need to talk to a doctor if you do not feel better or if you feel worse.
2. What you need to know before using Celluvisc
Do not use Celluvisc
- if you are allergic to caramel sodium or any of the other ingredients of this medicine (listed in section 6).
Warnings and cautions
- To avoid contamination or possible eye damage, do not touch the eye or any other surface with the tip of the single-dose container.
- Stop using Celluvisc and consult a doctor if you get eye pain, vision changes, redness, eye irritation, or if the eye gets worse.
Other medicines and Celluvisc
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
If you are using other eye drops , wait at least 5 minutes before using Celluvisc.
Pregnancy and breastfeeding
Celluvisc can be used during pregnancy and lactation.
Driving and using machines
Transient blurred vision may occur. You should not drive or use machines until you see clearly again.
You are responsible for assessing whether you are fit to drive a motor vehicle or perform work that requires increased vigilance. One of the factors that can affect your ability in these respects is the use of drugs due to their effects and / or side effects . Descriptions of these effects and side effects can be found in other sections. Read all the information in this leaflet for guidance. If you are not sure, talk to your doctor or pharmacist.
3. How to use Celluvisc
Always use this medicine exactly as described in this leaflet or as your doctor, pharmacist or nurse has told you. Ask your doctor or pharmacist if you are unsure.
The recommended dose is 1-2 drops in each eye if needed unless a doctor has given another prescription.
Instructions for use
To avoid contamination or possible eye damage, do not allow the open end of the single-dose container to come in contact with the eye or anything else. Wash hands before use.
1. Check that the single-dose container is intact
2. Break off the wing
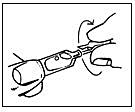
3. Take your drops as directed. Blink a few times.
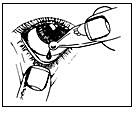
The opened single-dose container is used immediately and any remaining solution is discarded. It is very important that you throw away the solution and do not keep it.
A single-dose container is sufficient to treat both eyes. Celluvisc does not discolor contact lenses.
If you use more Celuvisc than you should
Accidental overdose poses no risks.
If you forget to use Celluvisc
Use the next dose as needed or at the next normal time as directed by your pharmacist or doctor. Do not take a double dose to make up for a forgotten dose .
If you have any further questions on the use of this product, ask your doctor, pharmacist or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects , although not everybody gets them.
Common side effects (affects more than 1 user in 100):
Eye irritation (including burning sensation and discomfort).
Has been reported (occurs in unknown number of users):
Burning, allergic reactions (including swelling of the eyelids or eyelids), sticky eyes, eye pain, itching of the eyes, var and / or drug residues in the eyelid edge, a feeling of having debris in the eye, redness of the eye / eyelid, blurred vision and / or impaired vision, eye damage to the surface of the eye due to the tip of the pipette touching the eye during use.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly to the Medical Products Agency, www.lakemedelsverket.se. By reporting side effects, you can help increase drug safety information.
5. How to store Celluvisc
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the single-dose container flap, foil bag, and carton after EXP. dat. The expiration date is the last day of the specified month.
Store the single-dose containers in the bag and put the bag back in the carton. The bag is needed to protect against evaporation of moisture. Do not store above 25 ° C.
Do not use this medicine if the Celluvisc carton does not appear to be untouched. Do not use Celluvisc if the solution changes color or becomes cloudy.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the packaging and other information
Content declaration
- The active substance is carmellose sodium 10 mg/ml
- The other ingredients are calcium chloride dihydrate, sodium chloride, sodium lactate, potassium chloride, and sterile water.
What the medicine looks like and contents of the pack
Celluvisc is a clear, colorless to pale yellow eye drop solution in clear endospipets . The single-dose container has a lid that is unscrewed. Each single-dose container contains 0.4 ml of eye drop solution.
Each package contains 30 or 90 single-dose containers in foil bags.
Each foil bag contains 10 single-dose containers .
Not all pack sizes may be marketed.
Marketing Authorization Holder and Manufacturer
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
County Mayo
Ireland