5 micrograms or 40 micrograms powder and solvent for injection , solution
alprostadil
1. What Caverject is and what it is used for
The active substance in Caverject is alprostadil . Alprostadil is a substance that is found naturally in the body and is also called prostaglandin E1. It dilates the blood vessels so that the blood flow in the penis is improved. This facilitates the onset of an erection .
Caverject is for the treatment of “erectile dysfunction”, which includes both weak erections and impotence . Caverject can also be used with other diagnostic tools when your doctor wants to find the exact cause of your erectile dysfunction.
2. What you need to know before using Caverject
Do not use Caverject
- if you are allergic to alprostadil or any of the other ingredients of this medicine (listed in section 6)
- if you have any disease that may increase the risk of prolonged erection , e.g. certain blood disorders such as sickle cell anemia, leukemia or bone marrow cancer (multiple myeloma )
- if you have an implant in your penis
- if you have a disease that causes you to avoid sexual activity (eg a serious heart disease)
- if you have a penis that is crooked or contains scar tissue
Do not give to infants, see information on benzyl alcohol in the section “Caverject contains sodium and benzyl alcohol”.
Warnings and cautions
- Painful erections can occur especially in patients with angular misalignment and foreskin constriction.
- Patients who have heart disease or lung disease as well as patients who have had a transient ischemic attack ( TIA , also called mini- stroke – a rapidly transient temporary lack of oxygen in any of the brain’s blood vessels) should use Caverject with caution.
- Patients with narrowing of the carotid artery or hypotension should use the drug with caution.
- If an erection lasts longer than 4 hours, a doctor should be contacted as soon as possible for any counter-treatment and discussion about dosage .
- Caverject does not protect against sexually transmitted diseases such as AIDS , herpes or gonorrhea . When injecting , a small amount of bleeding may occur at the injection site. This may involve an increased risk of transmitting such diseases. Condoms protect against sexually transmitted diseases.
- The reconstituted Caverject injection solution is for single use only. Any residue in the syringe should be discarded (see section 5. How to store Caverject).
- If the needle intended for injection is bent, change the needle to a new sterileneedle. Do not attempt to straighten a curved needle (see section 3 How to use Caverject).
Talk to your doctor or pharmacist before using Caverject if you:
- have one or more risk factors affecting the heart and blood vessels (for example, high blood pressure , tobacco use, high blood sugar, high cholesterol, overweight or obesity)
- have one or more risk factors for stroke (for example, high blood pressure , high cholesterol, coronary heart disease, irregular heartbeat or diabetes )
Children
Caverject is not intended for use in children. See also information on benzyl alcohol in the section “Caverject contains sodium and benzyl alcohol”.
Other drugs and Caverject
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
Do not use other medicines to treat erectile dysfunction at the same time as Caverject.
Tell your doctor if you are taking blood-thinning medicines, so-called anticoagulants , as such medicines may increase the risk of bleeding at the injection site.
Pregnancy, breastfeeding and fertility
Not relevant.
Driving and using machines
Caverject is not expected to have any effect on the ability to drive and use machines.
Caverject contains sodium and benzyl alcohol
This medicine contains less than 1 mmol sodium (23 mg) per dose , ie essentially ‘sodium-free’.
This medicine also contains benzyl alcohol. The preservative benzyl alcohol has been associated with serious side effects in children. The minimum amount of benzyl alcohol that can cause poisoning is not known. Caverject is not intended for use in children.
3. How to use Caverject
Always use Caverject exactly as your doctor has told you. If you are not sure, talk to your doctor or pharmacist.
The first injection is given by your doctor or nurse. Do not try to inject yourself until you have received thorough training and instruction from your doctor or nurse. The following instructions are intended as guidelines; if you have received other instructions, follow them.
If you want your partner to help prepare and inject Caverject, you should talk to your doctor or nurse so that your partner can also get instructions on how to proceed.
Dose one is determined by the doctor who adjusts it individually for you. The starting dose can be 2.5 micrograms or 5 micrograms, but you may need to increase the doseone to get the effect. Caverject should not be used more than once a day or three times a week.
The tables below describe the volume of solution to be injected depending on the dose prescribed by your doctor. The tables are divided by strength of Caverject.
Caverject 5 micrograms / syringe
| Prescribed dose | 5 micrograms | 3.75 micrograms | 2.5 micrograms | 1.25 micrograms |
| Volume to inject | 1 ml | 0.75 ml | 0.5 ml | 0.25 ml |
Caverject 40 micrograms / syringe
| Prescribed dose | 40 micrograms | 30 micrograms | 20 micrograms | 10 micrograms |
| Volume to inject | 1 ml | 0.75 ml | 0.5 ml | 0.25 ml |
To mix with Caverject:
- Wash your hands with soap and water.
- Open the package. Remove the plastic cap from the bottle of Caverject powder. Wipe the rubber stopper with one of the alcohol swabs.
- Open the package to the larger needle (size 22G with gray end). Leave the needle cover for as long as possible.
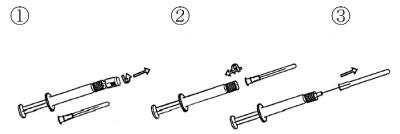
- Unscrew the seal from the spray tip.
- Press and tighten the sterile needle.
- Remove the needle cover. The syringe is now ready for use in reconstitution .
- Spray out any excess liquid. There should be 1 ml left.
- Insert the needle perpendicularly through the center of the bottle’s rubber stopper (see image below). Press the plunger down so that the liquid is injected into the bottle with the powder. Pull out the needle and spray out of the bottle. Then put on the needle cover and keep the syringe and needle in the package until you take your injection .
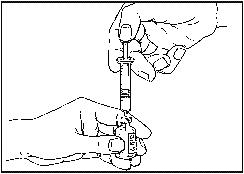
- Turn the bottle over a few times and let it stand upside down for a while so that the powder dissolves completely. Do not shake the bottle. The solution should be clear and colorless. Do not use the solution if it is cloudy, discolored or contains powder residues.
Increase the correct dose:
- Make sure that the plunger is completely depressed in the syringe. Insert the needle perpendicularly through the center of the rubber stopper of the bottle.
- Hold the bottle upside down (with the syringe underneath) and slowly inhale the amount of solution prescribed by your doctor.
- Check that there are no air bubbles in the syringe. These can be removed by tapping the syringe lightly with your finger or by slowly spraying the solution back into the bottle and then withdrawing the correct amount of solution again.
Replacement of injection needle:
- Carefully twist the larger injection needle from the syringe. The needle used should be disposed of (see section 5. How to store Caverject).
- Open the package for the smaller needle (size 30G with yellow end). Leave the needle cover for as long as possible.
- Press and turn the needle onto the syringe.
- Remove the needle cover. The syringe is now ready to be used for injection into the penis. To avoid breaking the needle, DO NOT use the needle if it is bent. Do not attempt to straighten a curved needle. If the needle is bent, replace the bent needle with a new, sterile needle.
Injection instructions
- Inject according to the doctor’s instructions with the syringe provided .
- Choose which side of the penis you want to inject. An alternate use of the pages is recommended. At the selected injection site, there should be no visible blood vessels and it should be in the middle of the penis side.
- Clean the injection site with disinfectant. Grasp the head of the penis and pull the penis close to the thigh.
- Hold the syringe in a straight line, alternatively at 45º, at an angle to the longitudinal direction of the penis and with a quick movement insert the needle completely into the penis (just under 1 cm).
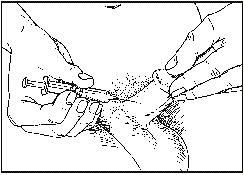
- Once you have inserted the needle correctly, inject the ENTIRE contents of the syringe for 3-5 seconds.
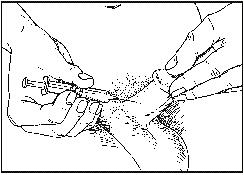
- DO NOT return any solution to the container. The needle and syringe used should be disposed of (see section 5. How to store Caverject).
- If necessary, press against the injection site for half a minute. If it starts to bleed, press until the bleeding stops. Depending on the size of the dose, it may feel burning in the penis in connection with injection one, then massage the injection site gently.
- If an erection lasts longer than 4 hours, contact a doctor immediately.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Very common (affects more than 1 user in 10)
- pain in the penis
Common (affects more than 1 user in 100)
- problems in the penis, scarring in the penis that can lead to calluses or that the penis becomes crooked
- prolonged erection
- bruise at the injection site. This should occur less frequently when you become accustomed to injecting
- reddening of the skin
- muscle cramps
Uncommon (affects less than 1 user in 100) are as follows
- reactions at the injection site may occur, e.g. the area may become inflamed, hot or swollen; it may itch, show rash or bleed
- the penis, testicles or scrotum may become red, swollen or sore, or the penis may become numb or extra sensitive, the ejaculation may change, the foreskin may feel tight, or you may have ollonitis; dilation of the seminal vesicle or various types of testicular disorders may occur
- Urinary incontinence may occur more often, it may feel uncomfortable to urinate or difficulty urinating may occur.
- if Caverject is accidentally injected into the urethra, small amounts of blood may appear in the urine or at the tip of the penis
- changes in blood pressure , increase in certain blood levels, palpitations and general weakness may occur as a result of the injection situation itself
- dizziness, pelvic pain, stiffness, nausea and dry mouth may occur.
- skin rash, increased sweating, extra heartbeat, fungal infection and cold symptoms have been reported
- increased or decreased touch sensitivity
- enlarged pupil er
- painful erection
- weak erection
- lowered blood pressure
- prolonged erection which lasts for a long time and which is often painful (priapism). Contact a doctor as soon as possible if you get an erection that lasts more than four hours
Has been reported (occurs in an unknown number of users)
- Insufficient blood flow to the heart muscle via the coronary arteries
- Stroke
The preservative (benzyl alcohol) can cause hypersensitivity reactions .
If you get any side effects, talk to your doctor or pharmacist. This also applies to any side effects not mentioned in this information.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist, or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly to the Medical Products Agency, www.lakemedelsverket.se. By reporting side effects, you can help increase drug safety information.
5. How to store Caverject
5 micrograms injection:
No special storage instructions. Do not use this medicine after the expiry date which is stated on the carton.
40 micrograms injection:
Do not use this medicine before the expiry date which is stated on the carton when stored at 2 ° C – 8 ° C (in a refrigerator) or within 3 months in the case of storage below 25 ° C.
Ready-made solution:
5 and 40 micrograms solution for injection: Shelf life for 24 hours not above 25 ° C. Do not freeze.
Keep out of sight and reach of children.
Do not use this medicine after the expiry date which is stated on the carton after EXP. The expiration date is the last day of the specified month.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the packaging and other information
Content declaration
- The active substance is alprostadil .
- Other ingredients:- 5 and 40 micrograms injection ingredient: lactose monohydrate , sodium citrate, hydrochloric acid , sodium hydroxide, benzyl alcohol, water for injections.
What the medicine looks like and the contents of the pack
5 and 40 micrograms of injection substance:
1 bottle of lyophilised powder and 1 pre-filled syringe with liquid for injection .
In addition, each pack contains 2 injection needles (22G and 30G) and 2 alcohol swabs.
The larger needle size 22G (gray end) is intended for reconstitution of solution and the smaller needle size 30G (yellow end) is intended for injection into the penis.
The bottle is of type I glass and is closed with a stopper of bromobutyl rubber and covered with an aluminum seal and a cap of polypropylene with flip-off function.
The syringe is of type I flint glass and is closed with a stopper and cap of butyl rubber.
Not all strengths may be marketed.
Marketing Authorization Holder and Manufacturer
Pfizer AB
191 90 Sollentuna
Tel: 08-550 520 00
Email: eumedinfo@pfizer.com