10 mg / ml injection solution
apomorphine hydrochloride
1. What APO-go PEN is and what it is used for
APO-go Pen contains apomorphine solution for injection. It is injected under the skin ( subcutaneously ). The active substance in APO-go Pen is apomorphine hydrochloride. Each milliliter solution contains 10 mg of apomorphine hydrochloride.
Apomorphine hydrochloride belongs to a group of medicines called dopamine agonists. APO-go Pen is used to treat Parkinson’s disease. Apomorphine reduces the time with “off” or immobile periods in people previously treated for Parkinson’s disease with levodopa and/or other dopamine agonists. Your doctor or nurse will help you recognize the signs of when you need to use the medicine.
Despite the name, apomorphine does not contain morphine.
2. What you need to know before using APO-go PEN
Do not use the APO-go PEN
Before using the APO-go Pen, your doctor will take an ECG ( electrocardiogram ) and ask for a list of all the medicines you are taking. The ECG examination will be repeated during the first days of treatment and at those times when the doctor deems it necessary. He or she will also ask if you have any other illnesses, especially with regard to the heart. Some questions and examinations may be repeated at each doctor’s visit. If you get symptoms that may come from the heart, e.g. palpitations, fainting, or fainting, tell your doctor immediately. You should also tell your doctor if you get diarrhea or start using new medicine.
Do not use APO-go PEN:
- you are under 18 years old
- you have difficulty breathing
- you have dementia or Alzheimer’s disease
- you have mental problems with symptoms such as hallucinations, delusions, confusion, lost perception of reality
- you have liver problems
- you have severe dyskinesia (involuntary movements), or severe dystonia (inability to move) despite treatment with levodopa
- you are allergic to apomorphine or any of the other ingredients of this medicine (listed in section 6)
- you or someone in your family has an abnormal electrocardiogram ( ECG ), which is called “long QT syndrome”.
Warnings and cautions
Talk to your doctor, pharmacist or nurse before using the APO-go Pen if:
- you have kidney problems
- you have lung problems
- you have heart problems
- you have low blood pressure or feel that you are about to faint or become dizzy when you get up
- you are taking any medicine for high blood pressure
- you suffer from nausea or vomiting
- you have some mental problems such as hallucinations and confusion due to Parkinson’s disease
- you are older or weak.
Tell your doctor if you or your family/carer notice that you develop a strong desire or desire to behave in ways that are unusual for you, or if you can not resist the impulse, drive, or temptation to perform certain activities. which may harm you or others. These behaviors are called disturbed impulse control and can include gambling addiction, compulsive eating, compulsive buying, an abnormally high sex drive, or an increase in sexual thoughts and feelings. Your doctor may need to adjust your dose or discontinue treatment.
Some patients experience addictive symptoms that lead to cravings for large doses of apomorphine and other medicines used to treat Parkinson’s disease.
Informant doctor or nurse if any of the above applies to you.
Children and young people
APO-go Pen should not be used in children and adolescents under 18 years of age.
Other medicines and APO-go PEN
Tell your doctor or pharmacist if you are taking, have recently taken, or might take any other medicines.
Talk to your doctor or pharmacist before using this medicine if:
- you use drugs that are known to affect how your heartbeats. This includes drugs used for heart rhythm problems (such as quinidine and amiodarone ), for depression (including tricyclic antidepressants such as amitriptyline and imipramine), and for bacterial infections (“macrolide antibiotics” such as erythromycin, azithromycin, and clarithromycin) and domperidone.
If you are taking this medicine while taking other medicines, it may be
- affect the effectiveness of these. This is especially true for:
- drugs such as clozapine used in the treatment of mental illness
- antihypertensive drugs
- other medicines for Parkinson’s disease.
Your doctor will tell you if you need to adjust the dose of apomorphine or any of the other medicines.
If you are taking both levodopa (another medicine for Parkinson’s disease ) and apomorphine, your doctor should take regular blood tests.
APO-go PEN with food and drink
Food and drink do not affect the effectiveness of this medicine.
Pregnancy and breastfeeding
The APO-go Pen should not be used during pregnancy unless clearly necessary.
Consult your doctor or pharmacist before using the APO-go Pen if you are pregnant, think you may be pregnant, or are planning to have a baby.
It is not known whether APO-go Pen is excreted in human milk. Talk to your doctor if you are breast-feeding or planning to breast-feed. Your doctor will explain to you whether you should continue/discontinue breast-feeding or continue/discontinue the medication.
Ask your doctor or pharmacist for advice before taking any medicine.
Driving and using machines
APO-go Pen can cause drowsiness and pronounced fatigue. Do not drive or use any tools or machines if this medicine affects you in this way.
You are responsible for assessing whether you are fit to drive a motor vehicle or perform work that requires increased vigilance. One of the factors that can affect your ability in these respects is the use of drugs due to their effects and/or side effects. Descriptions of these effects and side effects can be found in other sections. Read all the information in this leaflet for guidance. If you are not sure, talk to your doctor or pharmacist.
APO-go PEN contains sodium bisulfite
APO-go Pen contains sodium bisulfite which in rare cases can cause severe allergic reactions with symptoms such as rash, itching, difficulty breathing, swelling of the eyelids, face or lips, swelling, or red tongue. If you get any of these side effects, consult your nearest emergency department immediately.
APO-go Pen contains less than 1 mmol sodium (23 mg) per 10 ml, ie essentially ‘sodium-free’. is next to “sodium-free”.
3. How to use APO-go PEN
Before using the APO-go Pen, your doctor will make sure that you can tolerate this medicine as well as anti-nausea medicine, which you need to use at the same time.
Always use this medicine exactly as your doctor or pharmacist has told you. Ask your doctor or pharmacist if you are unsure.
Treatment with domperidone should be started at least 2 days before APO-go Pen treatment to prevent nausea or vomiting.
Do not use APO-go PEN if:
- the solution has turned green
- the solution is cloudy or if you see particles in it.
Where to inject APO-go PEN
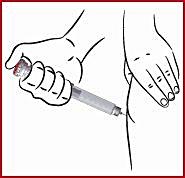
- inject APO-go Pen under the skin ( subcutaneously ), as your doctor or nurse has shown you
- APO-go PEN must not be injected into a vein.
Dosage
The amount and how often you need to use APO-go PEN is governed by your individual needs. Your doctor will discuss this with you and will tell you how much medicine to use. The dose that is suitable for you has been determined at an initial assessment at a specialist clinic.
- The usual daily dose is between 3 mg and 30 mg
- You may need up to 100 mg daily
- You usually need between 1 and 10 injections daily
- Each individual injection should not exceed 10 mg.
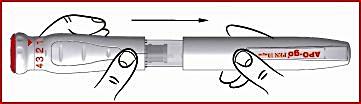
Before using the APO-go PEN, study your pen and the image below to familiarize yourself with your medicine.

* This package does NOT contain needles for pen use.
Use pen needles that are not longer than 12.7 mm (1/2 ”) and not narrower than 30G.
Pen needles recommended for use in insulin pens fit in the APO-go Pen.
IMPORTANT: Do not pull out the red dosing knob (see 1) before setting dose one (see “Setting the correct dose”).
Mounting the needle
(a) Before using the APO-go PEN, you will need sterile alcohol compresses and a needle in its protective shoe ( see 2 ).
(b) Remove the pen from the carton and remove the outer sleeve ( see 3 ).
(c) Wipe membrane ( see 4 ) with an alcohol swab.

(d) Remove the protective paper from the needle cone ( see 2 ).
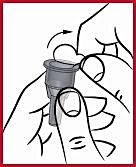
(e) It is important to keep the needle in line with the pen when attaching it, as shown in the illustration above. If the needle is not attached directly to the pen, the pen may leak.
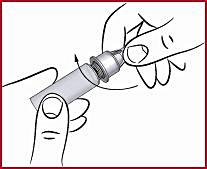
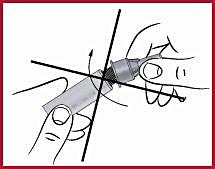
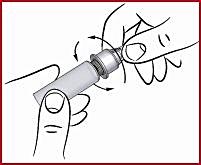
(f) Screw on the cone ( see 2 ) clockwise on the diaphragm until it is tight. This secures the needle securely.
(g) Remove the protective cone ( see 5 ), but do not throw it away. Do not remove the needle cover yet (see 6).
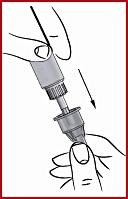
(h) Put the outer sleeve ( see 3 ) back on the pen.
Set the correct dose
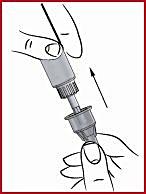
(i) Press down on the red dosing knob ( see 1 ) and turn the knob clockwise, while holding it down, until the arrow points to the prescribed dose ( see 7 and 8 ). Release the red dosing knob. Now the dose is set and you do not need to set the dose again before the subsequent injection.
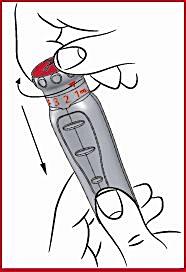
Important: If you happen to turn the steering wheel too far, just keep pushing down and turning the steering wheel in the same direction until the arrow points to the prescribed dose again.
Never turn and pull the red dosing knob at the same time.
If your dose is 1 mg, start by emptying a 1 mg dose on a paper handkerchief, for example, and discard it. This is called “preparing” the pen and is important because it ensures that you receive the full dose the first time you use your pen. Then set the desired dose for injection and inject as usual ( see “Injection” ). If your first dose is more than 1 mg, it is not necessary to prepare the pen.
Injection
(j) When setting dose one, carefully pull out the red dose knob as far as it will go. Check the red scale on the plunger ( see 9 ) and inject only if the barely visible line corresponds to the intended dose.
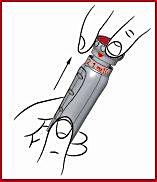

(k) Wash the injection site on the skin and around it with a sterile alcohol swab.
(l) Remove the outer sleeve of the pen ( see 3 ).
(m) Remove the needle guard ( see 6 ).
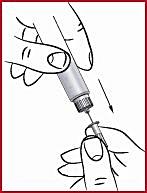
(n) Insert the needle ( see 10 ) as shown by your doctor or nurse.
(o) Inject by pushing down on the red dosing dial ( see 1 ) as far as it will go. Use your thumb if possible. When the red dosing knob is fully depressed, count to three before pulling out the needle.
(p) Attach the protective cone ( see 5 ) to the used needle and gently push it into place. When the cone is firmly in place, unscrew the needle counterclockwise. Dispose of the needle with the safety cone safely, in containers for used needles.
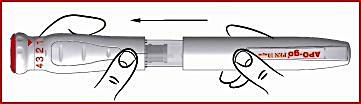
Preparation for the next injection
(q) Remove the outer sleeve of the pen and check that there is enough apomorphine in cartridge one for the next injection. If there is enough, fit a new needle in the same way as before.
(r) If there is not enough apomorphine in ampoule one for a new injection, prepare a new pen.
(s) Finally, replace the pen’s outer sleeve.
If you use more APO-go PEN than you should
- You may experience a slow heart rate, severe nausea, drowsiness, and/or difficulty breathing. You may also feel weak or dizzy, especially when getting up, due to low blood pressure. Lying down with your legs high makes you feel better.
If you forget to use APO-go PEN
Use the medicine the next time you need it. Do not use a double dose to make up for a forgotten dose.
If you stop using APO-go PEN
Do not stop treatment with APO-go PEN without first talking to a doctor.
If you have any further questions on the use of this product, ask your doctor, pharmacist, or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
If you get an allergic reaction, stop using APO-go PEN and consult a doctor or see the nearest emergency department immediately. Signs of an allergic reaction can be:
- rash
- breathing difficulties
- swelling of the face, lips, throat, or tongue.
APO-go PEN can sometimes cause the following:
Very common (may affect more than 1 user in 10):
- nodules under the skin at the injection site that are tender, painful, and maybe red and itchy. To avoid these knots, you should change the injection site each time you insert the needle.
- hallucinations (seeing, hearing, or feeling things that are not real).
Common (may affect up to 1 in 10 people):
- nausea or vomiting especially when you start using the APO-go Pen. If you are nauseous even though you are taking domperidone, or if you are not taking domperidone and are nauseous, talk to your doctor or nurse as soon as possible.
- fatigue or heavy sleepiness
- confusion or hallucinations
- yawn
- you feel dizzy or dizzy when you get up.
Uncommon (may affect up to 1 in 100 people):
- increased involuntary movements or aggravated tremors during so-called “on” periods
- anemia, abnormal breakdown of red blood cells in blood vessels or in other parts of the body. This is a less common side effect that may occur in patients who are also taking levodopa.
- sudden sleep attack
- rash
- breathing difficulties
- ulceration at the injection site
- decreased number of red blood cells, which can cause the skin to turn pale yellow and cause weakness and shortness of breath
- decreased platelet count, which may increase the risk of bleeding and bruising.
Rare (may affect up to 1 in 1,000 people):
- an allergic reaction
- eosinophilia, an abnormally high number of white blood cells in the blood or in tissue one.
Has been reported (occurs in an unknown number of users):
You may experience the following side effects:
- swelling of legs, feet, or fingers
- inability to resist an impulse to perform an action that may be harmful such as:
- a strong impulse to excessive gambling despite serious consequences for you personally or for your family
- altered or increased sexual interest or behavior that makes you or others noticeably concerned, e.g. an increased sexual drive
- uncontrollable and excessive need to buy things and spend money
- binge eating (eating large amounts of food in a short time) or compulsive eating (eating more food than normal and more than is needed to satisfy your hunger)
- fainting
- aggression, anxiety, restlessness
- headache.
Tell your doctor if you experience any of these behaviors to discuss ways to manage or reduce the symptoms.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist, or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly to the Medical Products Agency, www.lakemedelsverket.se. By reporting side effects, you can help increase drug safety information.
5. How to store the APO-go PEN
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the label and carton. The expiration date is the last day of the specified month.
Do not store above 25 ° C. Store in the original package. Sensitive to light.
The product must be stored under the same conditions after opening and between uses.
Do not use this medicine if the solution turns green. It should only be used if the solution is clear and colorless and free of visible particles.
When you start with a new APO-go PEN, this can be used for 48 hours. Do not reuse your APO-go PEN after this time. Take a new pen.
To dispose of your pen safely, always remove the needle from the pen and discard the needle in a container for cutting and stabbing objects. When the container is full, leave it at the pharmacy or discard it according to current instructions. Your used APO-go PEN must be handed in at the pharmacy.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the packaging and other information
Content declaration
The active substance is apomorphine hydrochloride. One milliliter of APO-go Pen contains 10 mg of apomorphine hydrochloride. An APO-go Pen contains a 3 ml solution for injection.
Other ingredients are:
sodium bisulfite (E222)
hydrochloric acid (37%)
water for injections
For information on sodium bisulfite, see section 2, “APO-go Pen contains sodium bisulfite”.
What the medicine looks like and the contents of the pack
APO-go PEN is a non-refillable pen that contains several doses. The pen contains a clear glass syringe with apomorphine solution for injection. The solution is clear, practically colorless, odorless, and free of visible particles.
Pack sizes
The package contains 1, 5, or 10 pens in a plastic tray in a cardboard box. Not all pack sizes may be marketed.
Marketing Authorization Holder and Manufacturer
STADA Arzneimittel AG
Stadastraße 2 – 18
61118 Bad Vilbel
Germany
Manufacturer
Britannia Pharmaceuticals Limited
200 Longwater Avenue
Green Park
Reading, Berkshire
RG2 6GP
UK
OR
Aguettant Laboratory
1 Rue Alexander Fleming
Mailbox 7144
69353 Lyon
Cedex 07
France
OR
STADA Arzneimittel AG
Stadastraße 2 – 18
61118 Bad Vilbel
Germany
Local representative
STADA Nordic ApS
Marielundvej 46A
DK-2730 Herlev
Denmark